CHEMISTRY IN CONTEXT// Chemical reactions and their implementation to detect alcohol levels in the human body

Author: @madridbg, through Power Point 2010, using public domain images.
Welcome back to all those readers of the #Hive community. This issue is intended to address the chemical reactions of oxidation and reduction, present in our daily lives, as well as those involved in determining the levels of alcohol in the human body through a technological instrument such as the breathalyzer. We will share the following guidelines from the @stemsocial community, which refer to scientific publications with practical contributions.
INTRODUCTION
The biological processes, are carried out mostly through aqueous medium, where a set of chemical reactions can be seen that allow the operation of the systems where they are developed. The term chemical reaction, has been coined in previous deliveries, where it is established as the process by which the reagents are transformed into products, giving rise to the formation of new substances with characteristics different from the original ones.
Therefore, on an experimental, biological and natural level, aqueous reactions play a preponderant role since they are present everywhere. On a practical level, reactions in the aqueous phase are very important, due to the capacity of water to dissolve a large number of substances, generating ionic substances that allow easy interaction and formation of chemical reactions.
In this sense, on this occasion we will focus on studying the set of reactions that can be carried out in the aqueous phase, concentrating on the redox reactions, in order to give an answer on the chemical functioning of the alcoholometer as an instrument used to avoid traffic accidents.
GENERALITIES OF THE SOLUTIONS IN AQUEOUS PHASE
As we have mentioned in previous deliveries, the dissolutions are homogeneous mixtures formed by a solute and a solvent, where the solute is the substance that is found in smaller quantity and the solvent is the most abundant compound within the solutions.
In function of instructing the reader on the behavior of the solutions, in this opportunity we will speak of electrolytic solutes, being understood as that substance that is completely dissociated in water, generated a solution and that has the capacity to conduct electricity. On the other hand, the non-electrolytic solutes, do not dissociate in water and therefore the solutions they generate do not have the capacity to conduct electricity.
A practical demonstration, to understand the phenomenon that takes place, consists of circulating electric current on different solutions and thus evaluate the capacity of dissociation of the substances involved, as shown in the following images.

Fig. 2. Equipment to identify electrolyte solutes. Author: @madridbg, through Power Point 2010.
Once the experiment is done we can determine that the capacity of a solution to conduct electricity depends on the amount of ions dissolved, so it is expected that sodium chloride (NaCl) is the compound that fully demonstrates this property, since it dissociates according to the following equation

Fig. 3. Chemical equation representing the aqueous dissociation of sodium chloride. Author: @madridbg, through Power Point 2010.
When analyzing the above equation, we realize that the positive and negative charges of the sodium and chlorine ions behave like an electrical bridge that allows the light bulb to turn on.
TYPES OF CHEMICAL REACTIONS
In this section of the topic, we will make a tour of the different chemical reactions that can develop in aqueous media, devoting greater attention to oxidation-reduction reactions (ORP) that interest us for practical purposes to comply with the title of this topic.
In this sense, we will make a brief outline of the reactions that take place in the aqueous phase.
Precipitation reaction: , Are those that result from the combination of certain cations and anions in solution, generating water-insoluble substances that tend to make solid, which are deposited at the bottom of the container because of its own weight. Precipitation reactions are used in laboratories to identify the ions present in a solution whose principle is based on the following equation.
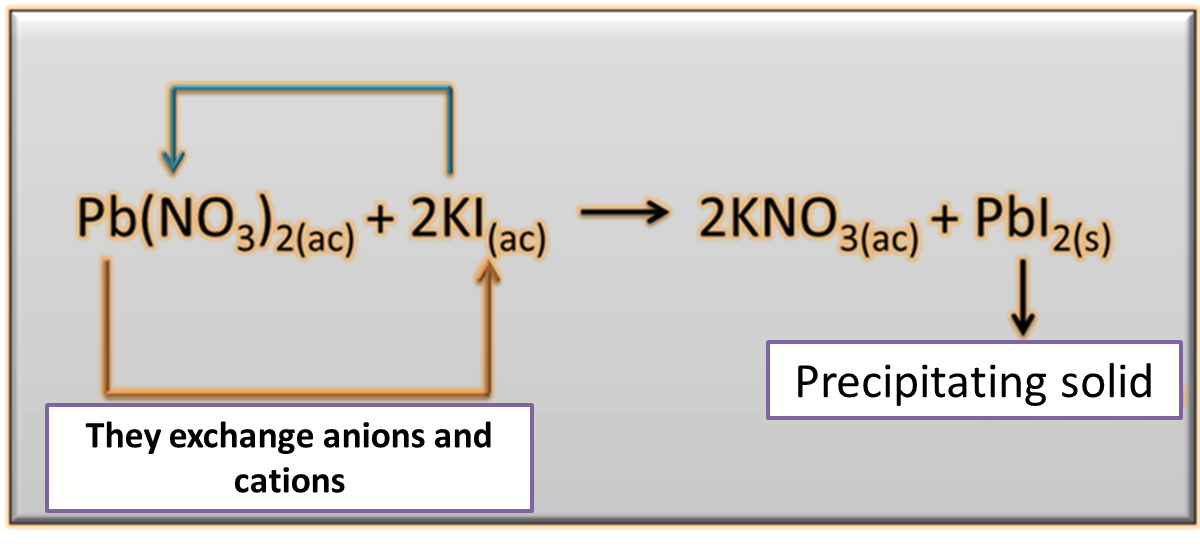
Fig. 4. Equation representing the precipitation of a chemical. Author: @madridbg, through Power Point 2010.
If we analyze the equation, we can see that combining an aqueous solution of lead nitrate [Pb(NO3)2] with a solution of potassium iodide (KI) results in potassium nitrate (KNO3) and the lead iodide (PbI2) that precipitates during the process, product of the double displacement of the cations and anions present in the reaction.
Acid-base reactions:
This type of reaction refers to the use of those substances that have the capacity to yield and accept electrons, which we call acids and bases respectively. When these substances react they give rise to the formation of a compound with ionic characteristics that we will call salt plus water production.

Fig 5. Equation representing an acid-base process. Author: @madridbg, through Power Point 2010.
If we analyze the equation, we can see that a hydrochloric acid (HCl) solution reacts as a sodium hydroxide (NaOH) solution resulting in the formation of sodium chloride (NaCl) plus water (H2O).
Reduction-oxidation reactions (ORP):
This type of reaction is produced as a consequence of the transfer of electrons from one substance to another within a solution, originating a variation in the charge of the atoms that participate.
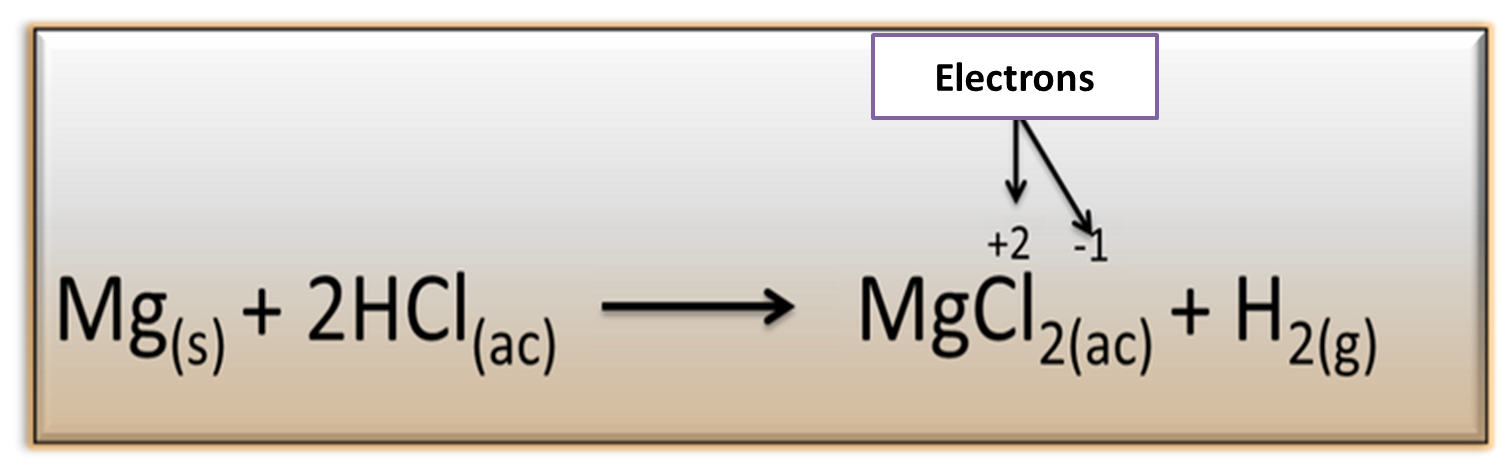
Fig 6. Equation representing an oxidation-reduction process. Author: @madridbg, through Power Point 2010.
An example of redox reactions is the interaction between magnesium (Mg) and hydrochloric acid (HCl), where magnesium yields one of its electrons to chlorine to produce magnesium chloride (MgCl2) and release hydrogen as a gas.
In order to understand the functioning of the alcoholometer, we will deepen in the redox reactions. Therefore, it is necessary to talk about the oxidation states of the atoms, which is defined as the number of charges that an atom would have in a molecule (or in an ionic compound) if the electrons were completely transferred. [1]
Through the number of oxidation we could evaluate if an element has oxidized or reduced, knowing that it has gained or lost electrons. In order to instruct the reader on redox reactions we will carry out a practical exercise, applying the concepts studied so far.
Proposed exercise:
The manganese oxide (MnO2) reacts with a solution of hydrochloric acid (HCl), resulting in the formation of manganese ions (Mn), chlorine gas (Cl2) and water, according to the following ionic equation, determine
1. Substance to be reduced
2. Oxidizing substance
3. Reducing agent
4. Oxidizing agent

Fig. 7. Chemical equation that represents the proposed statement. Author: @madridbg, through Power Point 2010.
The sequence to follow in order to respond to the exercise will be detailed in the following image.

Fig. 8. Execution of the proposed problem. Author: @madridbg, through Power Point 2010.
In the previous image we can observe that there are substances that oxidize and others that reduce, this according to the gain or loss of electrons as it was explained before, in the same way it is important to mention that in this type of reaction neutral compounds will always participate, which are present in the process but do not participate in the charge variation.
TYPES OF CHEMICAL REACTIONS
At the organic level, there is a great variety of compounds or organic family, in this respect we will talk about the alcohols, making reference to the ethanol that represents the fundamental principle of the alcoholic drinks. These substances have the capacity to oxidize and produce organic acids.
Therefore, when we are submitted to the use of the alcoholometer what is being measured is the amount of acetic acid that is formed as a result of the redox reaction between potassium dichromate and ethanol, present in our body because of the alcoholic beverages we consume.

Fig. 9. Oxidation-reduction reaction that takes place in the breathalyzer . Author: @madridbg, through Power Point 2010.
Technically, the device detects the loss of the orange color of the dichromate ion which turns green. This indicates that the chromium (+6) was reduced to chromium (+3) causing the color variation, so it is expected that the dichromate behaves as an oxidizing agent and ethanol will be our reducing agent.

Fig 10. Schematic representation of the operation of the breathalyser. Author: Chang, (2010). Edited by: @madridbg, through Power Point 2010.
The regulatory measures for considering an individual with ethanol intoxication depend on the laws of each country, however, a reference average that we can use is 0.1% blood alcohol per unit mass, which reflects the concentration of ethanol in the blood and per entity in our system.
CONTRIBUTIONS OF THE THEME
Through the themes of chemistry in context, we can integrate scientific content with daily applications of our daily lives. In this particular, the topic allowed us to approach those referring to the types of reactions, emphasizing those of oxidation and reduction and their application in instruments that allow us to watch over the integrity of human beings.
BIBLIOGRAPHY CONSULTED
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] Martinez and Col. (2018). The Breathalyzer: Alcohol Consumption in the National Fair of San Marcos 2018. International Journal of Educational Research and Training. Article: Online Access
[3] Mathews, C. K.; Van Holde, K. E.; Ahern, K. G. (2002) . Bioquímica. PEARSON EDUCACIÓN, S. A., Madrid, ISBN: 978-84-832-2694-0. Materia: Bioquímica, 577. ÁREA: CIENCIAS
[4] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[5] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[6] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCATION, MEXICO, 2011 ISBN: 978-607-32.793-5.
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/BGMadrid/status/1348443427940294658
https://twitter.com/BGMadrid/status/1348444386632355841
#posh twitter
https://twitter.com/BGMadrid/status/1348444386632355841?s=20
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.