CHEMISTRY IN CONTEXT// Basic oxides, their formulation and nomenclature.

Author: @madridbg, through Power Point 2010, using public domain images.
Welcome to all those readers of the #Hive community who are passionate about scientific content and especially to the members of the #stemsocial community who are at the forefront of educational topics. In this issue, we will be making a descriptive tour on the behavior of metal oxides in our society and we will focus on deepening the formulation and nomenclature of their formulation and nomenclature in an educational, entertaining and understandable way for the reader.
INTRODUCTION
The planet Earth is made up of a set of substances, some of which are combined, such as water, and others of invariable composition, such as iron. When these invariable substances are combined they form a great variety of compounds that we can find in the different states of matter.
This represents a determining factor in the development of life on the planet, to mention some related aspects, the human being uses the air, a mixture of oxygen and nitrogen, the rocks, a mixture of different minerals, the soil formed by different substances that allow the production and propagation of plants.
In this sense, there are many chemical compounds that can be found in nature, so it is necessary to have a control in order to assign a name to these substances according to their properties. That is why the chemical substances, which have been studied to date, are governed by a pattern established by the International Union of Pure and Applied Chemistry (IUPAC), which is the governing body that governs the rules for formulating and naming the more than 30 million chemical compounds that are known today.
The main function of this organism is based on establishing a standardized method that does not give rise to confusion in terms of compounds, where people can assign the same name to the substance, regardless of the country or language where they are. In other words, each name refers to a single species.
Therefore, in this publication we will study basic oxides, also known as metal oxides, which represent a group of compounds formed by the reaction of a metallic element with oxygen.
BASIC OXIDES GENERALITIES
Basic oxides, also called metal oxides, are binary chemical compounds formed by the combination of a metallic element with oxygen. They have the property of reacting with water to form ternary compounds which we will call hydroxides.
Oxides are very common in nature, product of the large number of metallic elements that we can find, likewise have the property of being good conductors of electricity and heat, also manifest high melting points and tend to make solids, although the properties of the substances from a combination of elements are different from the initial, such compounds usually have characteristics related to the element that originated them.

Fig. 1. Formulation of basic oxides. Author: @madridbg. Edited using Power Point 2010.
APPLICATIONS OF BASIC OXIDES
The usefulness and applications of oxides are related to the abundance of these compounds, due to the ability of metallic elements to combine with oxygen. In this sense, these compounds have a variety of uses of which we will describe some below:
- Zinc oxide (ZnO), a binary compound used in the manufacture of talc.
- Titanium dioxide (TiO2), used in beauty products, especially in makeup or cosmetics.
- Magnesium oxide (MgO), involved in acid-base reactions in antacids.
- Aluminum oxide (Al2O3), used in the manufacture of cans for carbonated beverages.
- Iron oxide (Fe2O3), used in the manufacture of alloys.
These are some of the uses, which have been given to the basic oxides in our daily life, which reflects the importance of knowing the formulation and nomenclature of these substances.
METAL OXIDE NOMENCLATURE
In this section, we will describe the procedure to follow to name the basic oxides according to the postulates made by the IUPAC. In this sense, it is necessary to mention that inorganic compounds can be named in correspondence to three types of nomenclature that we will describe below:
TRADITIONAL NOMENCLATURE (N.T)
This type of nomenclature focuses on the characteristics and oxidation states presented by the element that makes up the oxide, so it is necessary to describe a set of concepts to guide the reader on the use of this nomenclature.
To this effect, we should mention that a valence or oxidation states, is the capacity that an element has to combine and we represent them with positive and negative numbers, which tells us that the substance is accepting or giving up electrons.
Consequently, there are elements with only one oxidation state, which we will call monovalent, among which we can find sodium (Na=1+), aluminum (Al=3+), magnesium (Mg=2+), calcium (Ca=2+), lithium (Li=1+), among others.
On the other hand, the divalent elements, are those with two oxidation states, among which lead (Pb= 2+,4+), iron (Fe= 2+,3+), copper (Cu= 1+, 2+), nickel (Ni= 2+, 3+), among others, stand out.
Similarly, we find the polivalent elements, understood as those that exhibit three or more valences, here we can find chromium (Cr= 2+, 3+, 3+, 6+), manganese (Mn = 2+, 3+, 4+, 6+, 7+), among others.
Depending on the type of elements you are working with, you can follow some of the following procedures:
When the element is monovalent: Word oxide + de + metal name.
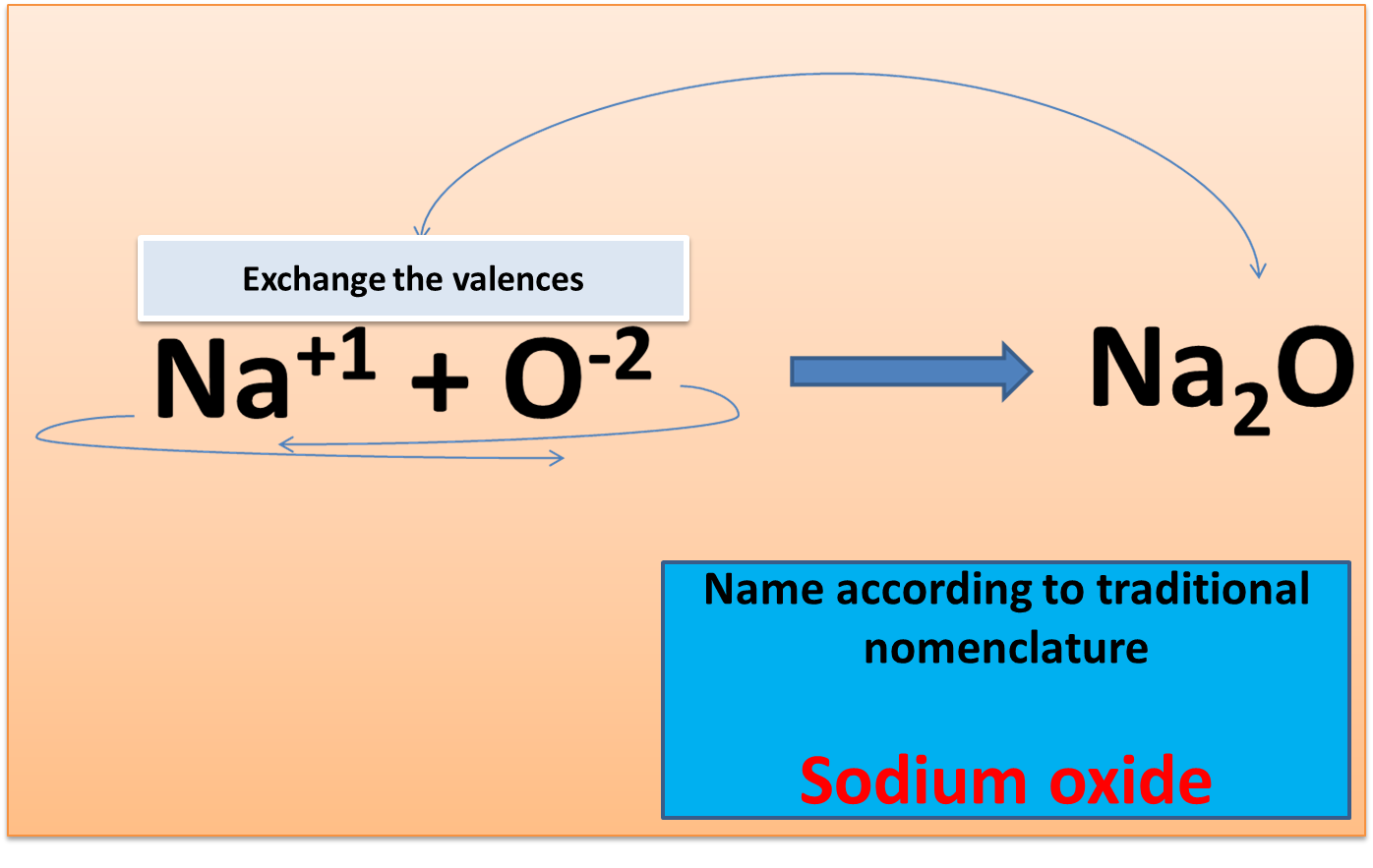
Fig. 2. Formulation and nomenclature of monovalent elements. Author: @madridbg. Edited using Power Point 2010
When the element is divalent: Word oxide + root of the metal + suffix in OSO and ICO.
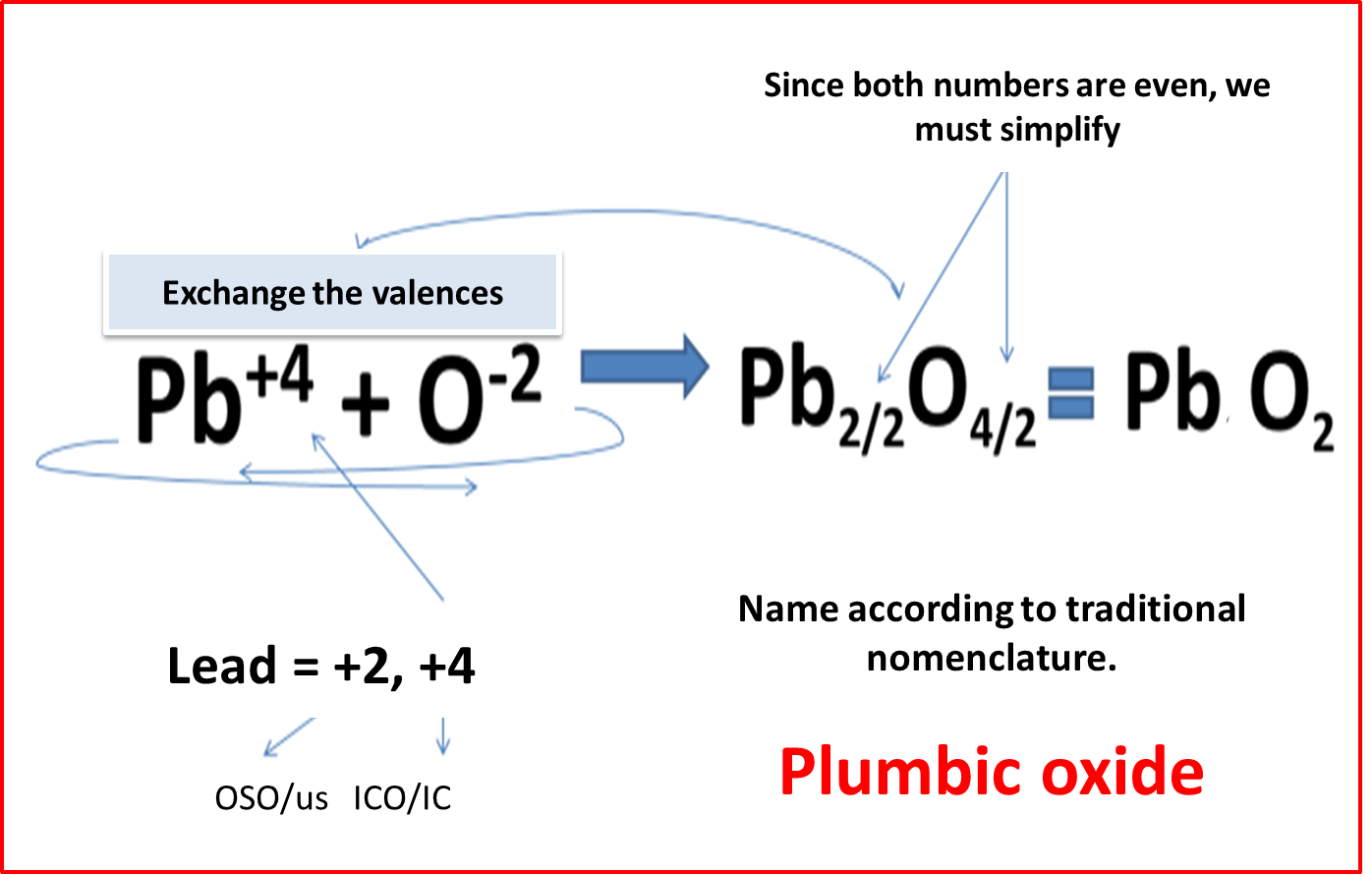
.Fig. 3. Formulation and nomenclature of divalent elements. Author: @madridbg. Edited through Power Point 2010.
When the element is polyvalent: Word oxide + prefix HYPO or HYPER + root of the metal + suffix OSO or ICO.
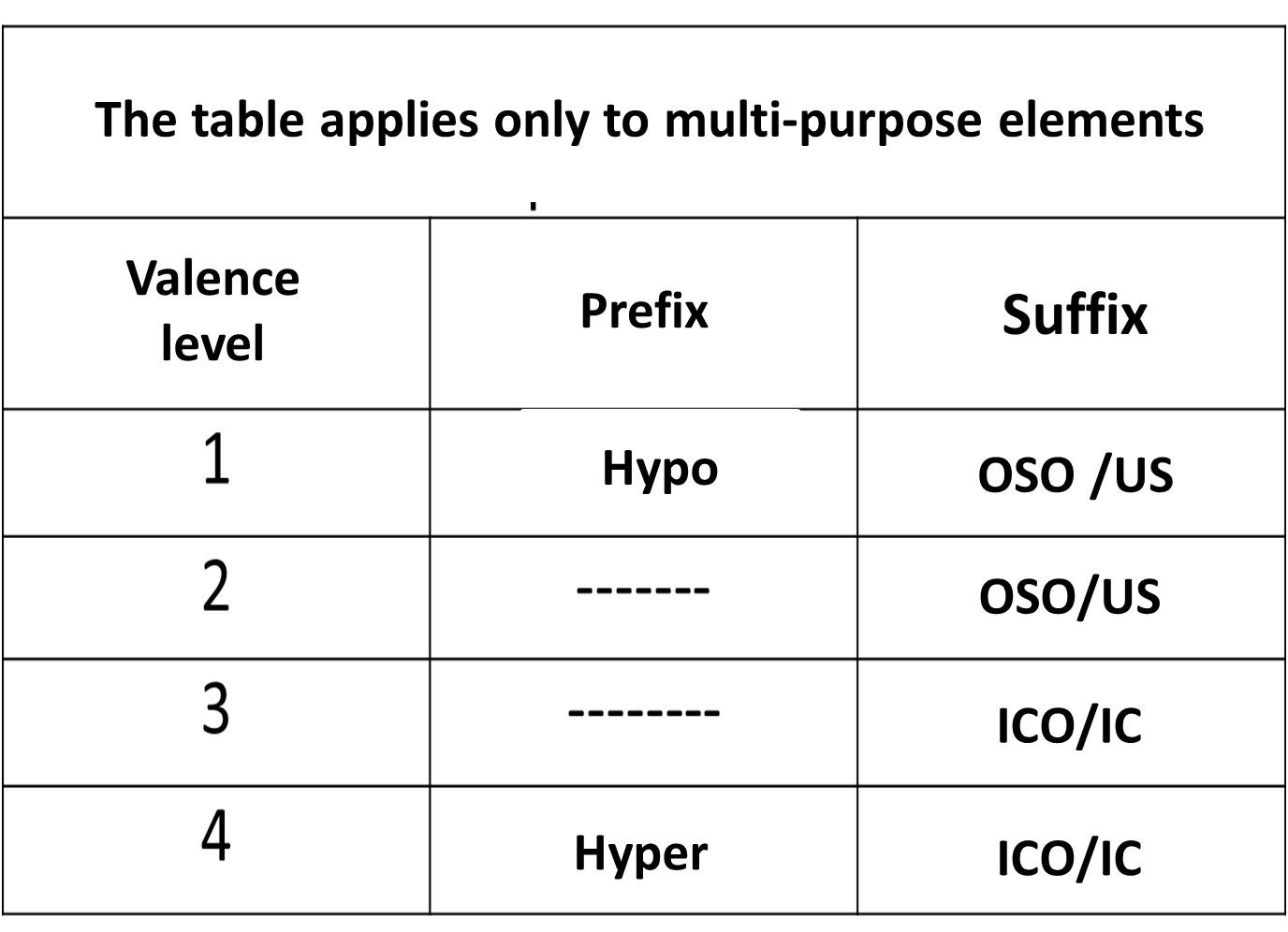
.Table 1. Formulation and nomenclature of polyvalent elements. Author: @madridbg. Edited through Power Point 2010.
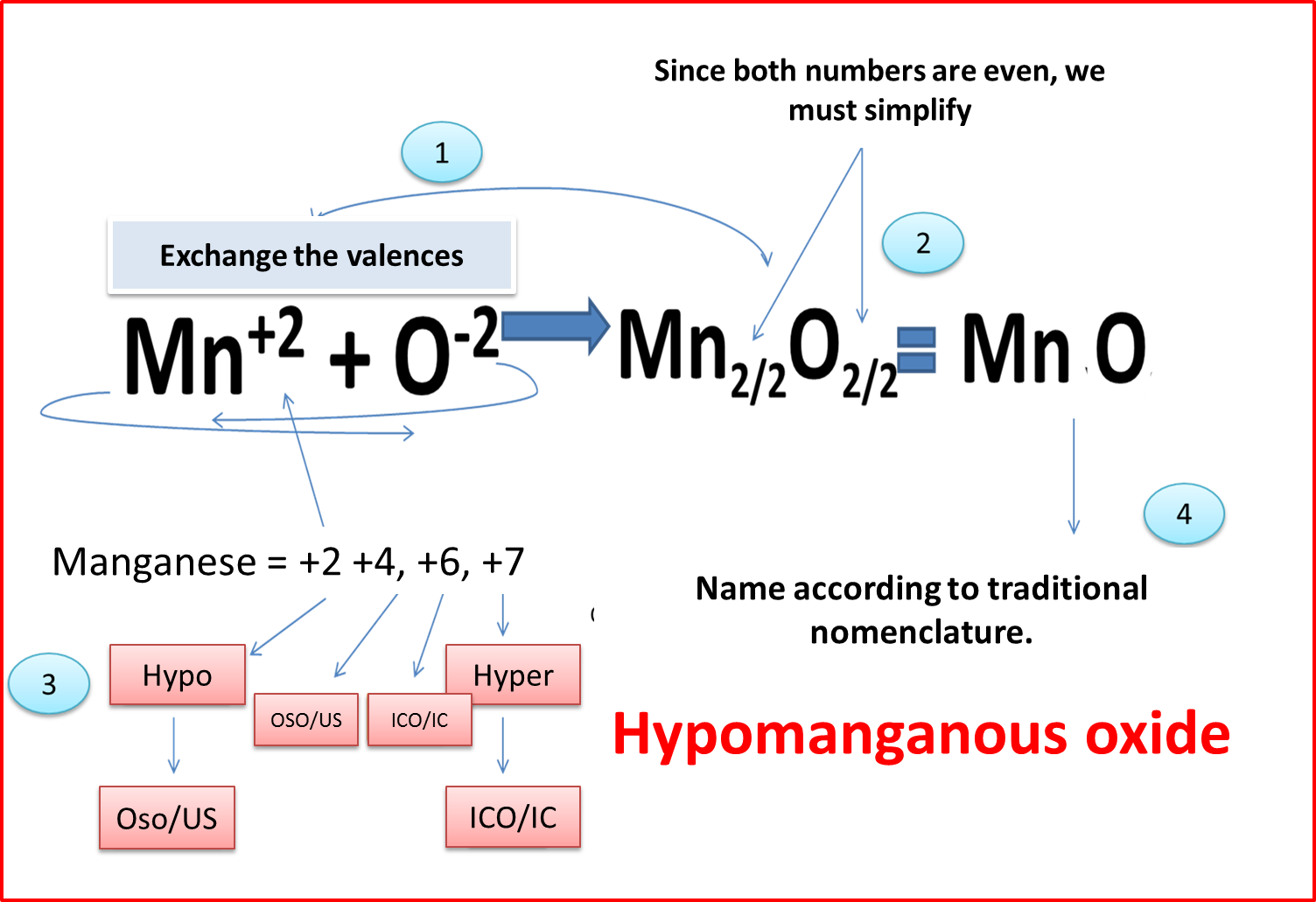
Figure 4. Formulation and nomenclature of polyvalent elements. Author: @madridbg. Edited through Power Point 2010.
STOCK NOMENCLATURE (N.S)
This is one of the simplest nomenclatures to work with and presents as variants the use of Roman numerals to identify the valency with which the metallic element is working, therefore only the following sequence is used.
Word oxide + de + metal name + valence in parentheses and in Roman numerals .
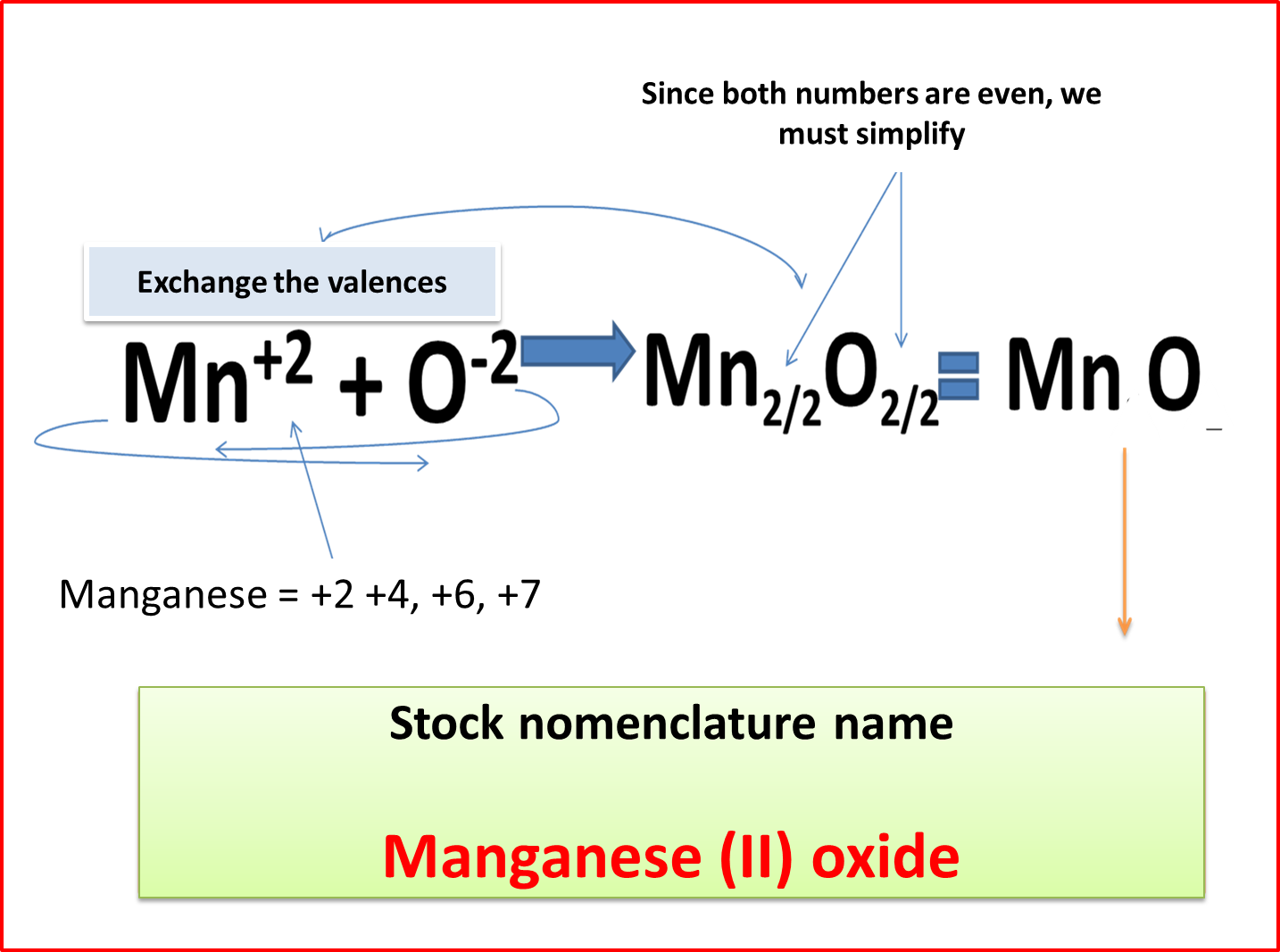
Figure 5. Stock nomenclature. Author: @madridbg . Edited through Power Point 2010.
MODERN NOMENCLATURE (N.M)
This nomenclature is based on the atoms that make up the compound, which are assigned Greek prefixes to identify them. As shown in the rules below.
Greek prefixes: mono (1), di (2), tri (3), etc. + word oxide + de + Greek prefixes + name of metal.
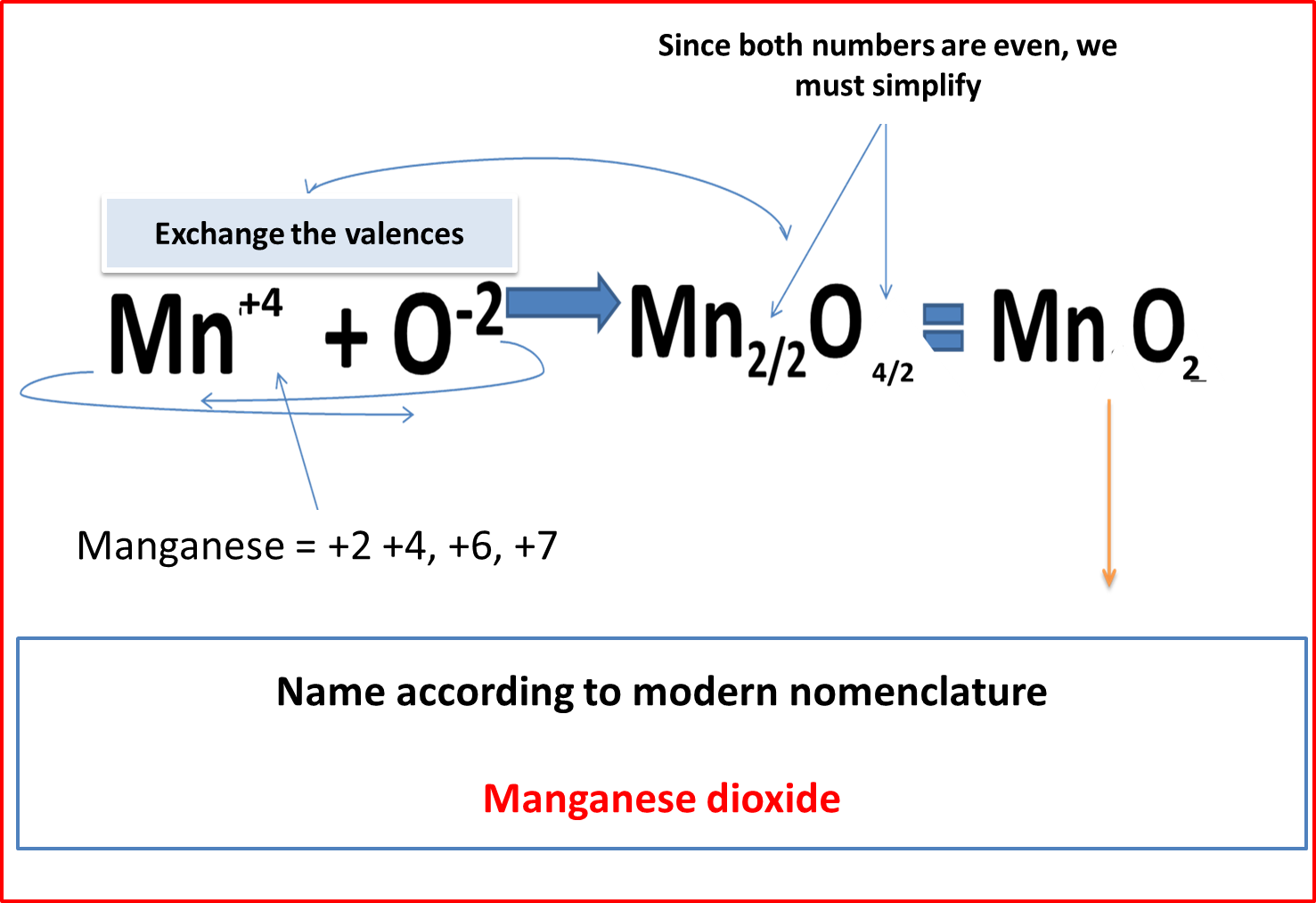
Fig. 6. Modern Nomenclature. Author: @madridbg. Edited through Power Point 2010.
In order to deepen the content addressed so far, we will attach an audiovisual material where the formulation and nomenclature of the basic oxides is developed through practical exercises.
Audiovisual material developed by. @madridbg. Edited through filmora software for 64 bit.
THEMATIC CONTRIBUTIONS
The descriptive material presented, allows transmitting a scientific content in a didactic way and adapted to the requirements of readers who are not trained in the area of science, specifically in chemistry. Likewise, it can be used as a methodological guide for teaching the formulation and nomenclature of inorganic compounds at different levels of education.
BIBLIOGRAPHY CONSULTED
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] Groel, N (2006). Generation of carbon dioxide by the reaction of an acid and a base Aquariological Silver Society . Article: Online Access
[3] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[4] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[5] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCATION, MEXICO, 2011 ISBN: 978-607-32.()793-5.
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/BGMadrid/status/1359616806651060234
#posh twitter
https://twitter.com/BGMadrid/status/1359616806651060234?s=20
Congratulations @madridbg! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s) :
Your next target is to reach 8000 upvotes.
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPCheck out the last post from @hivebuzz:
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.