Plastic bag walled saucepan for boiling water

Introduction
One of the phenomena that has benefited us the most throughout our history is fire, we have made it an essential part of our lives, and how not to do it, if it has allowed us to give the best use of our food, who has not enjoyed a tender roast or stewed meat, thanks to the action of heat generated by fire, or the direct action of it, many of the vegetables that we implement in our meals are edible due to the use of fire.
The use of fire has allowed us to fight diseases by eliminating parasites and bacteria from the food we eat, this to name just one essential example of the importance of fire in the life of mankind, by cooking food we spend less energy in the process of digestion, which benefits us in order to extend our longevity on earth.
All of us have been able to implement the fire or flame product of a combustion process, this process originated by the oxidation of those flammable materials when in contact with oxygen, therefore, the purpose of this article is to see if we can neutralize the degree of inflammation of a plastic bag, after being used as material for the walls of a small saucepan, where we will pour water to boil.
It is important to take into account that all flammable materials have the ability to emit gases into the atmosphere and, this is done at a regularly low temperature, these materials when mixed or in contact with a common oxidizer such as oxygen, causes and initiates the oxidation process that generates energy in the form of heat, such as fire.
Now, we have related in a general way with the fire and the process that generates it, where, we find as main element the inflammable material, the plastic bag is considered as one of these inflammable materials, therefore, it will be a challenge to boil water in our pan whose walls will be made of plastic bag.
We will see what happens with our small plastic-walled pan and also verify what other phenomena can help us to fulfill our purpose, so join me in this simple but interesting practical experience.
Making a small casserole with plastic bag sides
The implementation of physical science is possible in any space-time of our existence, that is why from my home I will share with you a simple practical experience, where, I will try to boil a portion of water in a pan made with plastic bag walls, for this, it is important that we know some of the materials to implement:
- Plastic bag.
- Soft wire.
- Liquid silicone.
- Water.
- Candle.
We will begin with the realization of our small casserole whose lining will be made of plastic bag, as you can see in the following images below:

After observing the previous image, I proceeded to start with the indicated purpose, that is, to try to boil water in our small pan lined with a plastic bag, this despite the fact that plastic is a highly flammable material when placed in direct contact with fire, let's see that this is true, in the following image.

In the previous figure, they could notice the high flammability capacity of the plastic bag when in contact with the heat generated by the candle or candle, "eye" highlighting that the plastic was not placed in direct contact with the candle, at a certain distance, and the plastic melted due to the effect of heat.
Now I proceeded to pour water into the small saucepan with plastic bag walls, with the help of a syringe I placed 10 ml of water in the small saucepan, and then placed it in contact with the candle.
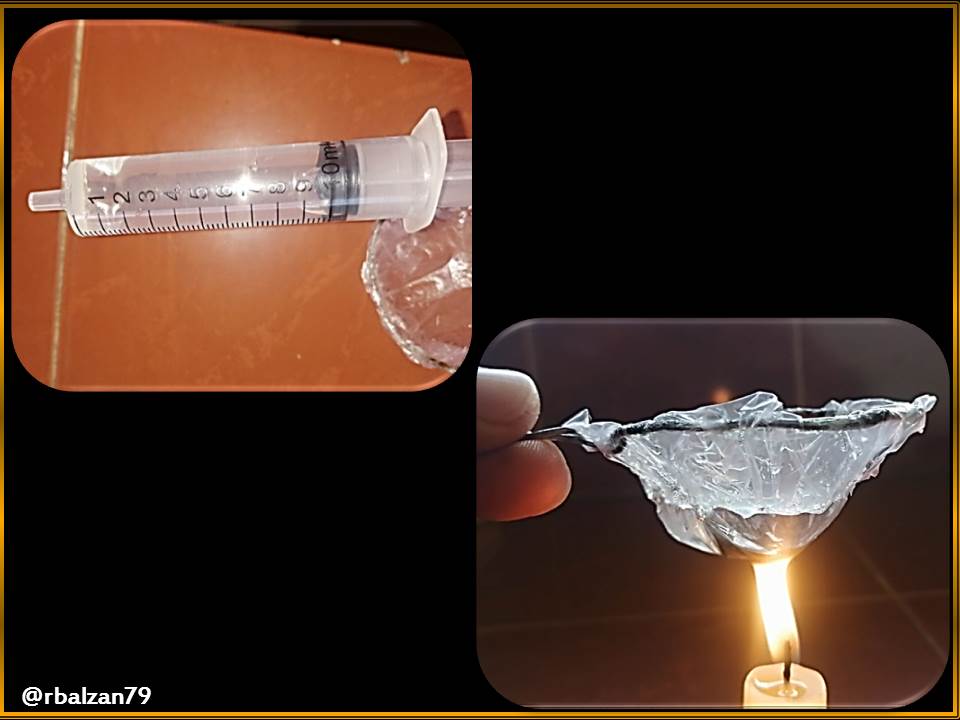
In this way to verify if it is possible to boil the water placed inside the pan, but after 2 minutes and 41 seconds the walls built with the plastic bag collapsed before boiling the water.

The heat supplied from the flame was being transferred to the water, therefore, the plastic did not melt instantly, as it did when we placed the pan without water, however, in this attempt we could not get the amount of water (10 ml) to boil.
This led me to perform a second test, but now, with 25 ml of water instead of 10 ml, and thus increase our volume of water, and thus, greater capacity for absorption of the heat of the candela, however, we must say that by increasing the volume of water, we will also require more time for this portion of water to boil.
Let's see what happens, and find out if now, this relationship of greater volume of water, makes us achieve the proposed purpose, for this, I show the following images.

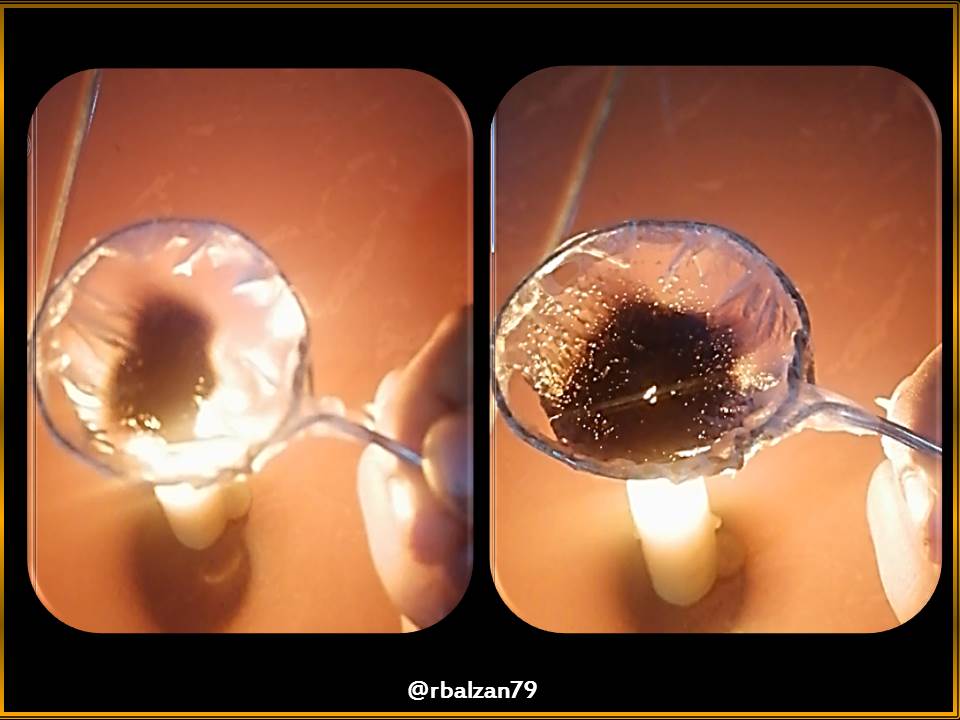
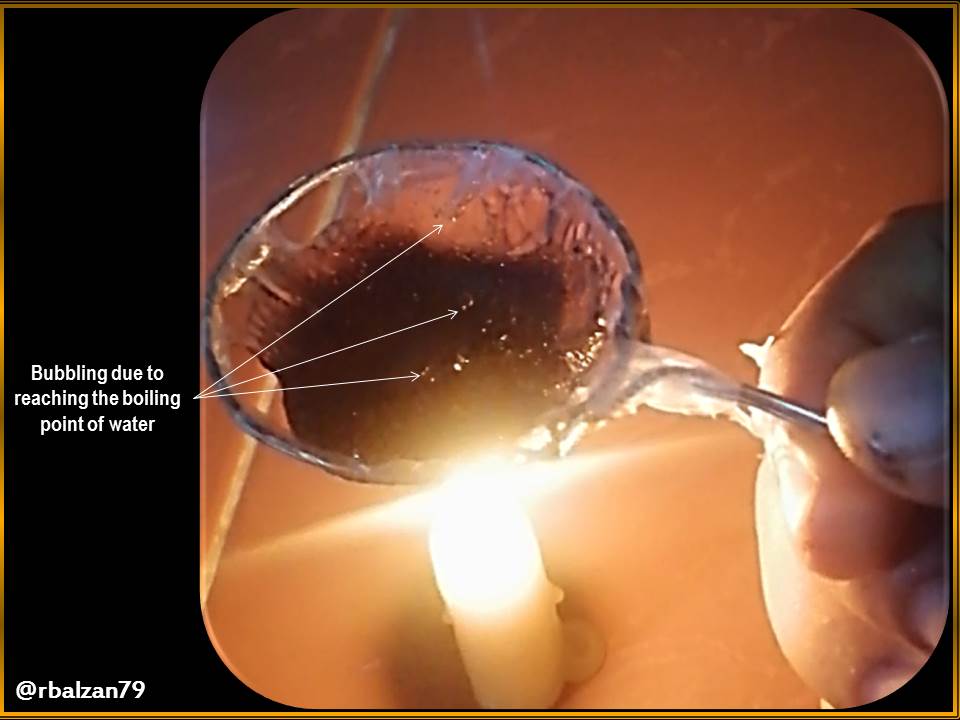
In the three previous figures we could see how the process was developing, where we noticed the presence of the boiling point of this volume of water, through the generation of bubbles, in this way the objective was achieved after a time of 5 minutes 05 seconds.
Then it is possible to heat the water to boiling, and without burning or melting the plastic bag, therefore, through the phenomenon of heat transfer, i.e., due to contact with water, the heat of the candela is transmitted from the plastic material to the water, this makes the temperature of the plastic bag walls does not reach its ignition level.
Conclusion
We are accustomed to use pans made of more resistant materials such as steel, aluminum, glass, among others, to boil any substance, such as water, now thanks to the heat transfer to the volume of water used, it was possible to boil the same, but, interestingly, using a small pan whose walls were built with very thin plastic bag.
They observed that the ignition temperature was reached when we placed our small pan in contact with the flame, and without water, also when we only poured 10 ml of water, however, in the second test with water the ignition temperature of the plastic was not reached, what we achieved was to reach the boiling temperature of water.
Without water, all the heat generated by the candle increases the internal energy of the plastic used, until it burns, as we noticed in the first two tests (one without water and the other with 10 ml), there are many phenomena that allow us to understand the action of our environment, as shown in this opportunity.
Note: All images are my own and worked in Power Point, and they were photographed with the camera of the HUAWEI Y9 cell phone, and the animated gif was made with Photo Scape.
Bibliographic references consulted and recommended
[1] Heat and mass transfer Link.
[2] Flammability of Polymers Link.
[3] CHEMISTRY AND PHYSICS OF FIRE Link.
esto me recordó algunos experimentos que realizaba en la educación de bachillerato, lo que hacía que las clases de física, química y biología se tornaran más interesantes.
Siempre usamos el fuego, desde época primitiva, pero nunca, o casi nunca nos detenemos a pensar en más allá. Buena muestra la que has realizado aquí, muy interesante.
Tienes toda la razón en lo que expresas, ya que cuando nos ponemos a analizar algunos fenómenos como el fuego, nos damos cuenta de su gran valor e influencia en la vida del ser humano. Gracias por tu visita a mi blog, y además, por tu valioso aporte. Saludos @apineda.