Lancet publishes a CDC study into the safety of the two mRNA COVID-19 vaccines in the US

In 2020 the US Food and Drug Administration (FDA) has issued an Emergency Use Authorization (EUA) for two mRNA-based vaccines against COVID-19: Pfizer's and BioNTech's BNT162b2 and Moderna's mRNA-1273. A recent study conducted by a CDC team and published in Lancet analyzes adverse effects caused by these vaccines.
Before we delve into further discussion it must be noted that the CDC is hardly an impartial entity. It is clearly an organization heavily involved in the promotion of COVID vaccines, including on social networks. So its organizational bias would likely be towards minimization of negative fallout from these vaccines. I think this helps to better appreciate the significance of this article.
The team behind the CDC study needs to be commended for providing at least some metrics corresponding to the adverse effects, including fatal ones, resulting from the vaccination campaign. That is a welcome contrast to the public narrative declaring these vaccines as "safe and effective", so much so that only a crazy "antivaxxer" would even consider the idea that they may be anything but.
Let us now look at this study. It covers the two out of three main vaccines used in the US: the two mRNA based vaccines. They do cover the majority of vaccine doses administered. But one must note that there is one more vaccine technology used, Janssen by Johnson and Johnson, a viral vector vaccine technology. And, unless we assume that Janssen is 100% safe, we need to accept that the data on the two mRNA-based vaccines does not cover all adverse effects from the COVID vaccination campaign in the US.
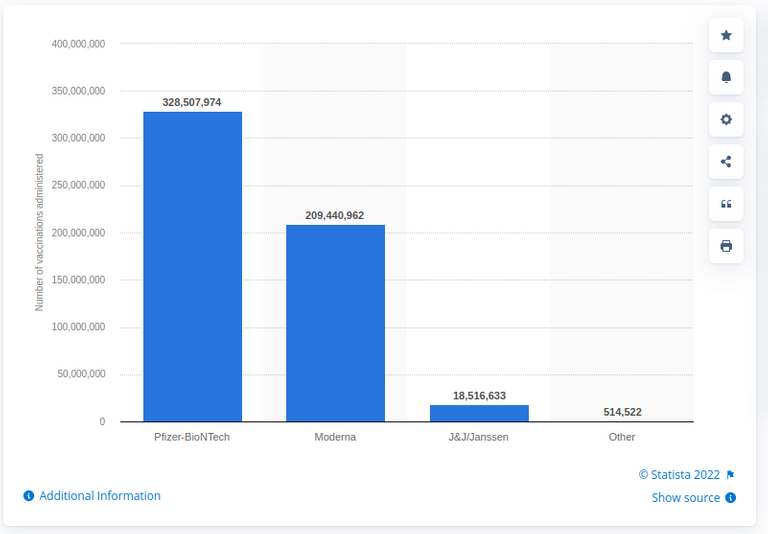
Image: Statista
The two mRNA vaccines are administered as a two dose sequence. The Janssen is a single dose. It is to be noted that neither the CDC study nor just about all other do not clearly correlate the number of doses and the number of vaccinated individuals. That introduces significant confusion into the study,
The interpretative conclusion of the study reads as follows, "Safety data from more than 298 million doses of mRNA COVID-19 vaccine administered in the first 6 months of the US vaccination programme show that most reported adverse events were mild and short in duration." This statement is not incorrect and the situation it signifies is medically banal as mild forms of many health disorders occur more frequently than severe ones. Thus, one may say, it may be fairly described as a fancy way of saying nothing, or nothing of interest.
To get at the meat of the study one needs to actually read the data in it. As stated above, the metric used is the number of doses administered, not the number of individuals vaccinated. Confusion caused by this ambiguity inevitably affects the whole analysis of the study downstream.
The time period covered by the study is 6 months from 14 December 2020 through 14 June 2021. During that time period, 298,792,852 doses of the two vaccines were administered. Two systems were used to track the outcomes, VAERS and V-Safe. VAERS is governmental vaccine effect tracking system run by the FDA and CDC. V-Safe is a CDC-managed self-reporting system based on a mobile app. That, I must note, makes it likely that a serious fatal event may not make it into V-Safe as most people choose not to use their mobile devices while unconscious or dead. V-Safe data is designed to trigger a follow up to VAERS when judged appropriate by medical professionals processing V-Safe reports. This transition also introduced potential for data loss or corruption.
For the purposes of this analysis we will only look at the two mRNA vaccines as an aggregate. This is just for the sake of brevity - you are obviously encouraged to look into it in more detail. So we have a total of nearly 300 million doses of the vaccines delivered. Assuming every recipient received the two doses mandated e have a total of roughly 150 million individuals vaccinated. The other extreme would have every recipient only receive one dose, and that gives us roughly 300 million recipients. The truth is obviously somewhere in between.
VAERS had collected a total of 340,522 reports. Of those, 313,499 (92.1%) were non-serious, 27,023 (7.9%) were serious and 4,496 (1.3%) were deaths. So let us consider this data in context of the overall vaccination programme. Assuming the highest estimate of individuals involved was 300 million. That gives us a minimum fatality rate of 14.9 per million.
That would be quite low at the first glance. However, vaccinations are medical interventions administered to healthy individuals and are thus expected to be supremely safe. For example, the 1976-1977 swine flu vaccination campaign was abandoned after several hundred cases of adverse effects, mostly Guillain–Barré syndrome, and about 25 fatalities in about 45 million vaccinated individuals. That gives us a fatality rate of 0.6 per million.
So in 1976 a mass vaccination campaign, which was not compulsory was abandoned due to a fatality rate of 0.6 per million while we have just had a mass vaccination campaign whose minimum fatality rate as reported by a CDC study published in a major scientific publication is 14.9 per million, or some 20 times higher. The silence that surrounds this clear discrepancy is both deafening and disturbing.
References
Safety of mRNA vaccines administered during the initial 6 months of the US COVID-19 vaccination programme: an observational study of reports to the Vaccine Adverse Event Reporting System and v-safe | Article PDF
Hannah G Rosenblum, MD, Julianne Gee, MPH, Ruiling Liu, PhD, Paige L Marquez, MSPH, Bicheng Zhang, MS, Penelope Strid, MPH, et al., The Lancet Infections Diseases, 7 March 2022
Number of COVID-19 vaccine doses administered in the United States as of March 13, 2022, by vaccine manufacturer
Statista, 13 March 2022
Moderna COVID-19 Vaccine: document re: FDA EUA Authorization
Moderna, 15 March 2022
FDA approves Pfizer/BioNTech coronavirus vaccine for emergency use in US
Jessica Glenza, The Guardian, 12 December 2020
CDC's vaccine promotion post on Facebook
CDC, 18 March 2022
1976 swine flu outbreak (wiki)
Archived 30 July 2021
Swine flu ‘debacle’ of 1976 is recalled
Shari Roan, Los Angeles Times, 27 April 2009
Johnson & Johnson’s Janssen COVID-19 Vaccine Overview and Safety
CDC, 22 February 2022
Social media links
Website
Support
View or trade
BEER.Hey @borepstein, here is a little bit of
BEERfrom @pixresteemer for you. Enjoy it!Learn how to earn FREE BEER each day by staking your
BEER.These are the things they're reporting first. Imagine what will be revealed over the next year. The silence of the people who called "conspiracy theorists" every time people tried to warn them speaks volumes.
This is by no means unique either. Gulf War syndrome is pretty recent and has ongoing effects. All for a vaccine to something that wasn't there. It's amazing how people forget these significant events and then repeat them.