Journal club - the anticipatory immune response of pain-sensing neurons in skin
Our body evolved an immune system to fight infections. Our bodies also evolved the feeling of pain to avoid injury and make us aware of the site of infection. But is it possible that the nerves which sense pain, also prepare distant sites in the body to prepare for impeding infections and injuries? Let's find out in today's journal club.

Image adapted from max pixel | CC0
The paper
The paper I am going to put up for discussion this week is titled - Cutaneous TRPV1+ Neurons Trigger Protective Innate Type 17 Anticipatory Immunity. It was published this month in Cell by Cohen et al., from Daniel Kaplan's lab.
What is so interesting about it? What is the take-home message?
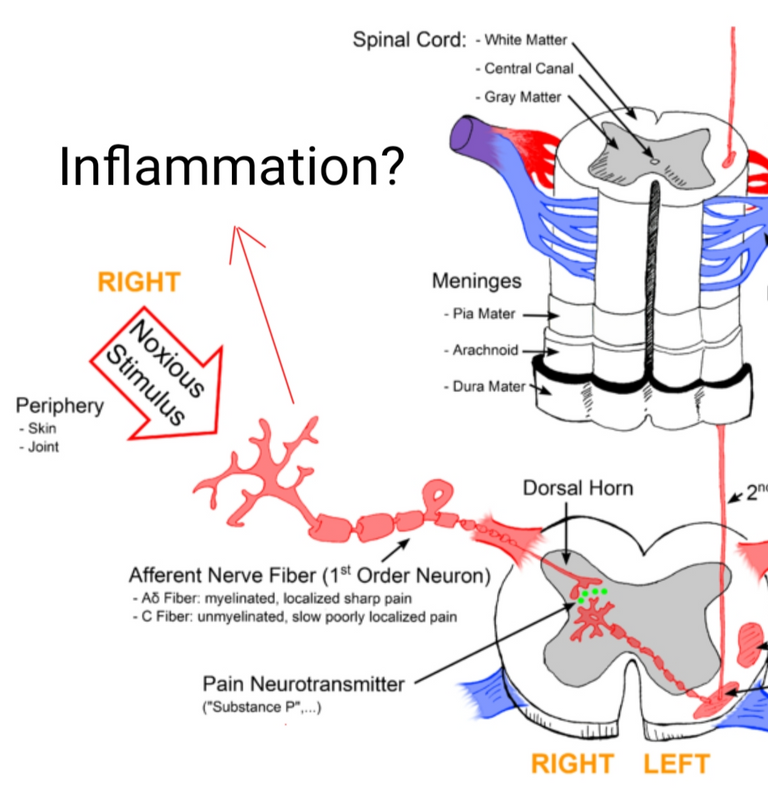
Image by Bettina Guebeli | CC BY-SA 4.0.
Well, before this paper we had some idea that the nerves in the skin, that play a role in pain sensation, also play a role in initiating an immune response to some pathogens. However, what was not known was if these nerves are sufficient in isolation to any other cell type to initiate the immune response. The authors say they are sufficient for kick-starting type 17 (aka type 3) immunity. Moreover, the paper also discovers a mechanism for anticipatory immunity. That means if you get an infection at one site in the skin the nervous system is capable of initiating an immune response at distant sites as well as if it is preparing the body for upcoming danger. Plus, they use a pretty cool technique to do so, called optogenetics, which I will talk about soon.
A tat bit of background
TRPV1+ afferent neurons
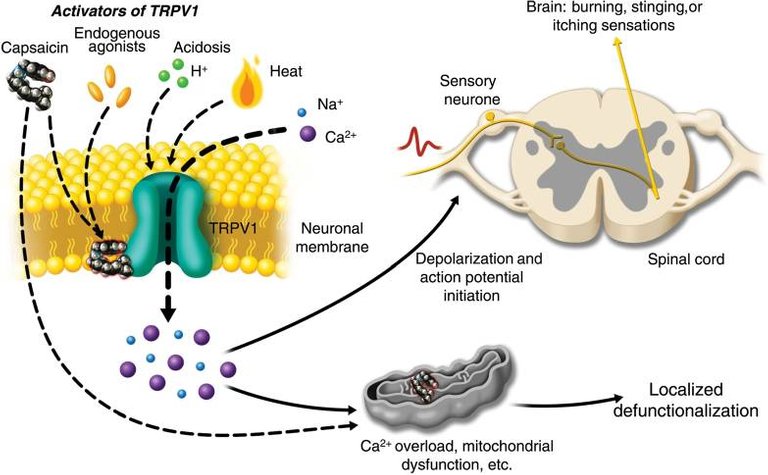
Image by Anand and Bley, 2011 | CC BY-SA 2.5.
TRPV1+ afferent neurons (transmit information from periphery to spinal cord) are a subset of nerve fibres that innervate the skin. Their major job profile over there is of the nociceptor, which are the neurons which sense pain. They were initially discovered as neurons which respond to capsaicin, the compound in chilli peppers such as capsicum. However, with time they have also been shown to convey the feeling of pain associated with heat, acidic pH, cellular damage related molecules and even pathogen-related molecules (Szallasi et al., 2007). They are also the neurons which makes you itch when your body senses an irritant (Kittaka et al., 2017). Moreover, these neurons when activated also secrete neuropeptides such as substance P and CGRP, which can cause inflammation (Roosterman et al., 2006). It has also been shown that chemically induced dermatitis (the itchy inflammation of skin), kind of a model for psoriasis, requires TRPV1+ neurons (Riol-Blanco et al., 2014).
Even though it has been shown that molecules from pathogens like - staphylococcus aureus, Candida and Streptococcus pyogenes, can activate TRPV1+ neurons and skin and induce type 17 immune response; it is not clear if these neurons are sufficient to explain the immune response or if they are just part of stuff that happens (Cohen et al., 2019).
A brief intro to types of immune responses
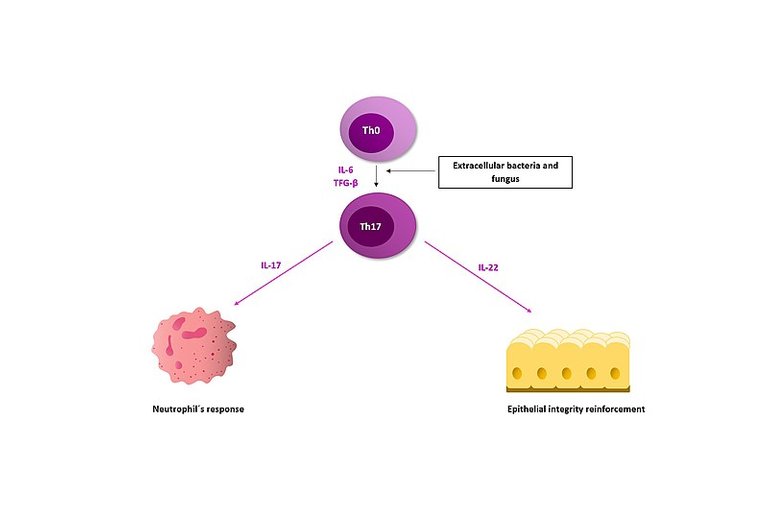
Image by Cecilia Tejero García | CC BY-SA 4.0.
The immune response of our body can be put in 3 broad categories type 1, type 2 and type 3 (aka type 17). They are classified based on subtypes of T cells and cytokines that are recruited in mediating the immune response. For instance, type 1 response is categorised by activation of T helper 1 (Th1), and T cytotoxic 1 cells, and natural killer cells which produce interferon-γ, TNF and LTa. These cytokines also activate phagocytic cells and cause B cells to produce IgG, IgM and IgA antibodies. Type 1 immunity hence helps the body to fight against intracellular pathogens. On the other hand, the type-2 immune response is devoted to fighting against worms, venom and other allergens. Type 2 immunity is characterised by the presence of Th2 and Tc2 cells, which produce IL4, IL5 and Il13. These cytokines cause B cells to produce IgE and recruit mast cells, eosinophils and basophils (Anunziato et al., 2015).
But, the one important to us for today is the third type of immune response aka type 17, is activated by extracellular by bacteria and fungi, such as those that live on the skin. It is activated by cytokines such as IL-23, TNFa, IL-6, IL1B. It is characterised by Th17, Tc17 and γδT-cells which produce 1L17 and IL22 as signature cytokines of type 3 immunity. These cytokines also target the epithelial the cells to produce anti-microbial peptides which target the bacteria on the skin. This is the kind of immune response that is linked to TRPV1+ cell activation. However, are these sensory neurons enough to kick start type 17 response to skin pathogens, is the question of the day.
So what's the story
The blues after adding channelrhodopsin to TRPV1+ neurons
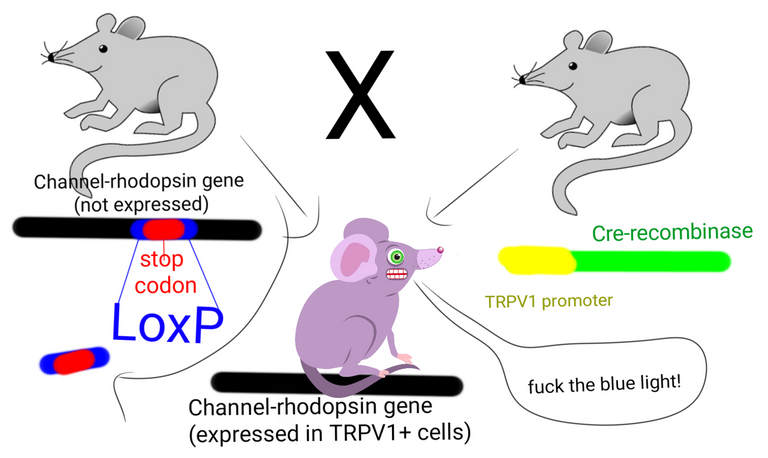
Illustrated by @scienceblocks using the following images -
Mouse cartoon 1 by da5id1 | public domain
Mouse cartoon 2 | Pixabay
Image by Millencolin | CC BY-SA 3.0
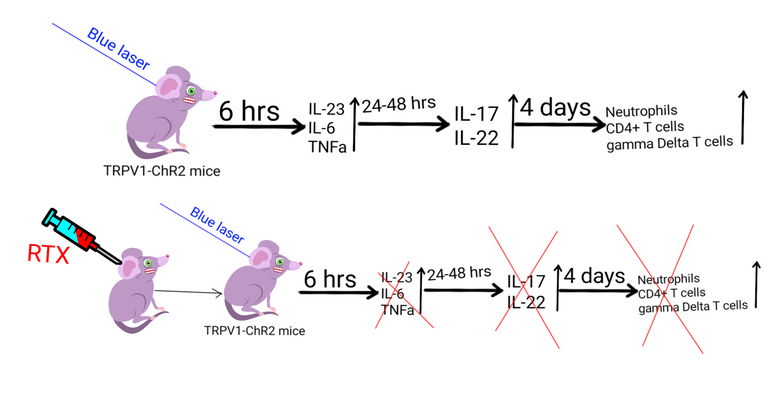
In order to test whether TRPV1+ neurons are sufficient to activate type 17 immunity, the authors used an optogenetic approach. Which means that they expressed this protein called channelrhodopsin specifically in TRPV1+ neurons of the mice. So now, these neurons can be activated just by shining blue light on them. Sadly, the first thing that happens when you shine blue light on one of the ears of these mice is that it feels pain. Also, when they did this for 30 mins per day for 5 days, the entire ear became thick, inflamed and displayed psoriasis-like features, indicative of type 17 inflammation.
Illustrated by @scienceblocks
Mouse cartoon 2 | Pixabay
Syringe by pitr | public domain
When they dissociated the ear and passed the cells through a flow cytometer, they could detect the presence of γδT-cells, T helper cells and neutrophils - characteristics Type 17 response. Moreover, the authors also observed that 6 hours after they said -"let there be light", there is also an increase in expression of inflammatory cytokines - IL-23, IL-6, TNFa - the activators of type 17 immunity. And then 48 hours later IL-17 and IL-22, the signature cytokines of type 17 immune response went up. However, none of this was observed in the ear not exposed to blue light or in control animals with channelrhodopsin exposed to light. Moreover, inhibiting TRPV1+ neurons with a chemical called RTX, also inhibited the inflammation of the ear and recruitment of immune cells.
Neuropeptide CGRP mediates the effect of TRPV1+ neurons.
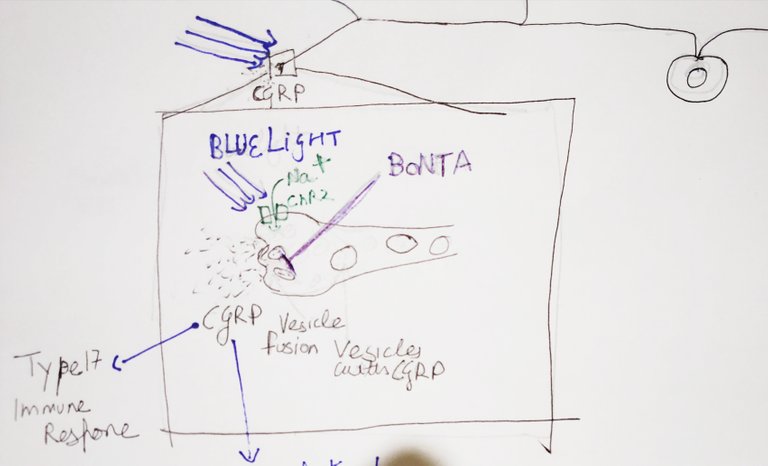
Doodled by @scienceblocks
Since, CGRP, the neuropeptide secreted by these neurons, also went up when the neurons saw the blue light, the authors wanted to know if it is required for mediating the immune response. To test this they injected botulinum neurotoxin A into the dermis of mice. This neurotoxin blocks the vesicle fusion in neurons which is required for neuropeptide secretion. In the mice injected with this neurotoxin and photostimulated with blue light, the expression of cytokines did not go up.
But why neurons?
Now given that TRPV1+ neurons can be directly activated by molecules of the pathogens, and the experiments above showing that they are sufficient to activate the immune response - it means that these neurons are part of the first line of defence against extracellular microbes (of course after the skin barrier). But why did neurons need to evolve something that could have been taken care of by other resident immune cells, or for that matter even by some cells that form the skin? Why in the freaking world neurons need to work extra hours for this?
If you think about it neurons can do something other cells don't. They can transmit the signal in a matter of milliseconds using electric impulses. They can even relay the information to the brain. In a way, they can warn the body about the infection (which they do by transmitting pain) much faster, and it is possible that they can even start an immune response at other places before the infection reaches there. So like when infection happens at one place, and then start spreading to a nearby place, the immune cells there would be like - "haha, we were expecting you". It would be an ambush. So does it happen?
Because they spread the message
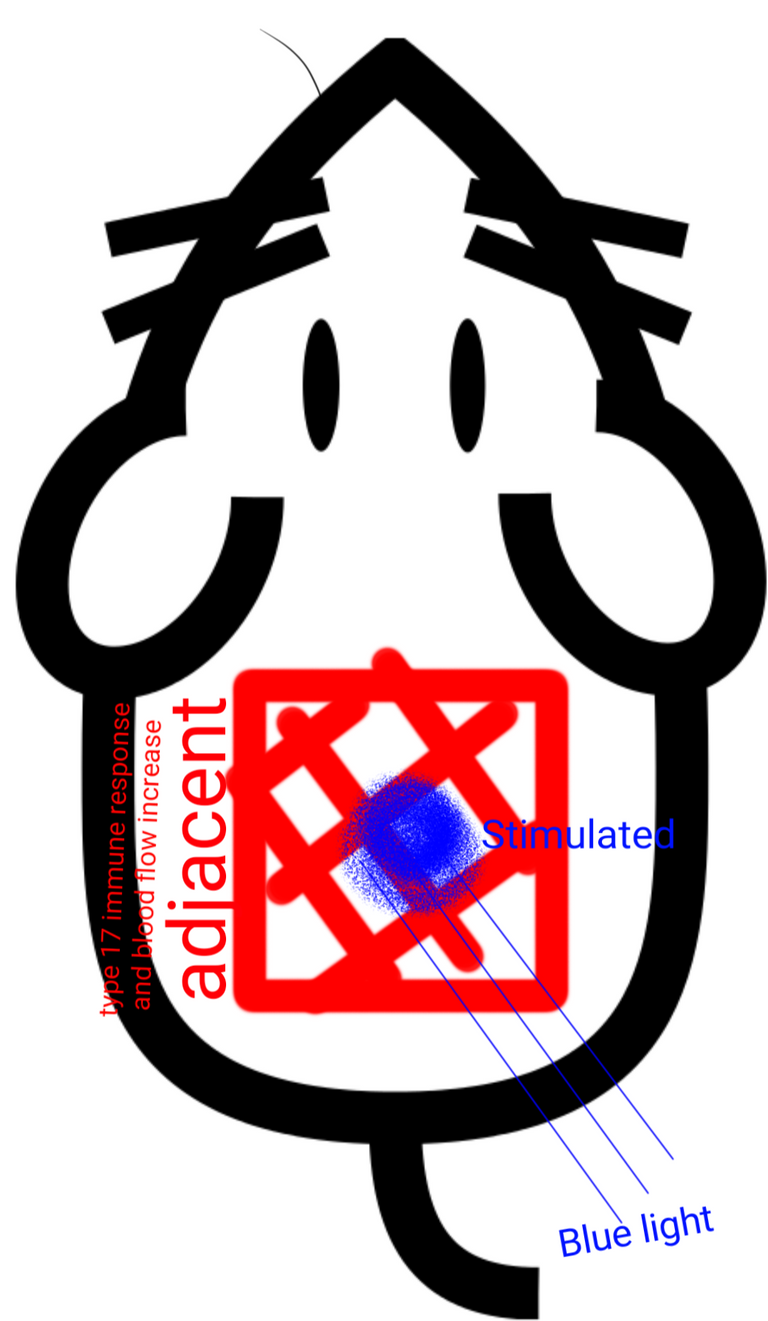
Adapted from mouse cartoon by Alanavc | CC BY-SA 3.0
One thing with the neurogenic inflammatory response is that it also increases the blood flow from nearby vessels. So to test if activation of these neurons also activated nerve fibres in nearby non stimulated regions, the researchers used 2D doppler ultrasound to measure the blood flow in different regions. They would shine blue light at one region in back skin and measure the blood flow in that area as well as nearby area. And, voila! The blood flow increased not only in the stimulated area but also in the adjacent non-stimulated area. They also found CGRP being released both at the site of stimulation and in nearby areas. The same help true for expression of inflammatory cytokines - IL-23, IL-6 and TNFa. Moreover, when they photostimulated the skin before infecting it with Candida, the Candida infection found it hard to spread to adjacent regions, in mice expressing channelrhodopsin vs control mice.
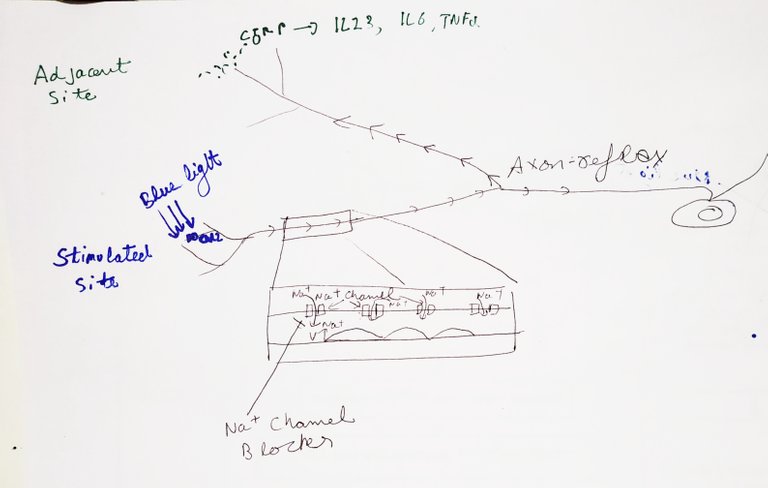
To test if it is indeed the neurons that are spreading the message via electrical impulses, they blocked the impulses using a drug called bupivacaine. Bupivacaine is a voltage-gated sodium channel blocker and is hence used to stop neuronal communication. When they injected bupivacaine in dermis before shining the blue light, the CGRP and inflammatory cytokines were released at the site of stimulation but not in nearby areas. Moreover, when they injected the botulinum neurotoxin A (to block CGRP neuropeptide release), they found that the inflammatory cytokine expression was only blocked at site of injection and stimulation but not in nearby areas. Confirming that the signal to activate the immune response in adjacent areas travel via electrical impulses.
Finally, to test if the neurons can also respond the same way to Candida, they infected one of skin site with heat-killed fungus (so that the fungus can't spread on its own but its molecules can activate the neurons). The heat-killed fungus kick-started the type 17 immune response both at the site of infection and in nearby areas. The immune response in nearby areas even this case could be blocked by bupivacaine. Moreover, if the mice pretreated with heat-killed fungus were now infected with live fungus, they live fungus found it hard to spread to adjacent areas. Well, except with these mice were also treated with bupivacaine.
These experiments make a strong case not only about TRPV1+ neurons being sufficient for kick-starting the immune response to pathogens but also for the warning the nearby areas to prepare for war. Of course like any other papers, there are things that one need to be careful about - for instance TRPV1+ expression is not only limited to neurons, it can also be expressed by other immune cells and epithelial cells. However, authors do address this and show that on their model the expression of channelrhodopsin was limited to neurons. However, why that might be the case in their model is not very clear. Then, in their model, the channelrhodopsin got expressed in both peptidergic and non-peptidergic neurons TRPV1+ neurons. Though they talk about this, it might need further investigation to confirm their results in a more specific model.
Nevertheless, their results are interesting not only in terms of anticipatory immunity to pathogens; but also makes you wonder about other scenarios such as wound healing. Like, can getting wounded at one site prepare you to respond faster to would at the adjacent site? How far and how fast can this immune response prepare the body for incoming pathogens and injuries? What if this system is activated without any reason at one site - say via tumour, can this also make inflammatory environment-friendly or detrimental to tumour metastasizing to another site. Can this system also explain the psoriasis flare-up by psychological stress (well because type 17 immunity is implicated in the pathology of psoriasis)?
A bit about journal club series
This is a new series I am starting. In this series rather than discussing an entire field say about diabetes, CRISPR or ageing, I will summarize that paper I liked a lot, and we can have a discussion on it. We often do this in the lab, so I thought why not on steemstem? In comments do let me know if you liked the idea and approach, and if you think I should add or change something. It would be nice of you to put up questions on the paper and fuel discussion. Also, if you like it, I would encourage you to write about your favourite papers.
About steemstem
But, before I go I would like to mention about the steemstem platform. Well, if you love reading and writing interesting science articles @steemstem is a community on steem that support authors and content creators in the STEM field. If you wish to support steemstem do see the links below.
You can vote for steemstem witness here -Quick link for voting for the SteemSTEM Witness(@stem.witness)
Quick delegation links for @steemstem
50SP | 100SP | 500SP | 1000SP | 5000SP | 10000SP).
Delegating to @steemstem gives an ROI of 65% of the curation rewards.
Also, if you have any questions regarding steemstem, do join the steemstem discord server.
You can DM me on discord, I have the same handle - @scienceblocks. Also if you are not a steem user, and reading this blog inspired you to start your science blog, find me on discord and let me know about you. I can try and help you navigate your way through steem.
Cohen et al., 2019. Cutaneous TRPV1+ Neurons Trigger Protective Innate Type 17 Anticipatory Immunity
Roosterman et al., 2006. Neuronal Control of Skin Function: The Skin as a Neuroimmunoendocrine Organ
Wow. Bookmarked and resteemed.
I had/ still have chronic pain.
It helped me a lot to realise that pain is just information! making progress :)
Im going to study ur post very intensely, now ;)
also the < under one of ur first pics, for the center code, is being shown.. ;)
Hey, thanks for point that out, fixed it. :)
Posted using Partiko Android
Interesting. Thanks!
Hi @scienceblocks!
Your post was upvoted by @steem-ua, new Steem dApp, using UserAuthority for algorithmic post curation!
Your UA account score is currently 3.615 which ranks you at #6114 across all Steem accounts.
Your rank has dropped 160 places in the last three days (old rank 5954).
In our last Algorithmic Curation Round, consisting of 164 contributions, your post is ranked at #116.
Evaluation of your UA score:
Feel free to join our @steem-ua Discord server
I finally printed your article out, took it aside and read it in a quiet place. This material is challenging for me, but still there was much to mine. I'm very interested in the immune response, which is really the heart of your article, especially immune responses that are generated far from the site of an event. Your speculation about psoriasis flares coincided exactly with where my mind was going as I finished your piece.
Systemic lupus came to mind. This disease is famously triggered by just about anything. An assault on the body sends signals throughout to go on high alert, and a flare, unrelated to the original event, will ensue. This is similar to a psoriasis flare. Of course, in lupus, while the original antagonist might not be dangerous, the ensuing flare can be.
Very perplexing for lupus patients and treating physicians. Whenever an assault on the body (or mind) occurs, there is always consideration of an indirectly related immune response.
While this paper was hard for me to work my way through, because the technical language is unfamiliar to me, still there was much value in reading.
Thank you for taking the time to write and explain these important matters. You turn SteemSTEM into a laboratory for learning--for scientists and non-scientists.
Thanks for this great feedback once again. That's is a very interesting connection you made there. There are not many studies on this, but there is some correlational evidence, in support of picture of SLE flare that you drew. The TRPV1+ neurons seem to be hyper-responsive in SLE. For instance, in this poster abstract the authors suggest that skin reaction to capsaicin (the chemical activator of TRPV1+ neurons) is much stronger in SLE patients. Of course we would need much more evidence than this, but I think its a promising direction to ponder in. Actually, this whole anticipatory immunity opens up a whole new pandora's box. For instance, we know chronic pain can be path to depression. We know depression is associated with low grade inflammation in body. Is there a connection between these two dots? I don't know. It's a far fetched hunch. But it's worth giving a shot to this experiment.
Also, my apologies that I got a bit carried away this time and didn't simplify the technical terms. I know I ended up talking as if I am presenting this in my lab. This feedback would be helpful when I write the next journal club post.
Don't modify your writing style because I find it challenging. My training is in history, humanities, languages and literature (yeah, I really sampled everything in the liberal arts!). Science is a new horizon for me. I love it, have always loved it, but lack some of the tools a trained scientist would have. Reading your articles, and others on SteemSTEM.io, helps me to learn. There's a saying that if you want to be a better chess players, then engage with better players.
Write for the community. I will wander around, sort of like an avid tourist, and learn from all of you as I go. It's an adventure. And, as with an aspiring chess player, I'll get better :))
No, I am not going to modify my writing style. More like improve it and leave no jargon is left unturned. The reason I love to write about science is so that information reaches everyone. Sometimes it does get difficult. For instance, that Rosa26 concept, for us at lab its an everyday thing. But while writing it I was unsure until the end, if the message would go across or not. It is something which may require a post of its own. The challenge is how to not omit it and yet make sure that a 12-year-old reading it would have all nice images popping in their head. I like knowing if I did a good job at that or not. Which is why I love feedback like this. I am not going to change my chess game, I am just going to open up more on why that certain move made sense to me. 🙂🙃
Posted using Partiko Android
😄

I look forward to learning from you, and to upping my chess game.
That journal club series is a great initiative. I hope you will get a lot of comments and questions. I am somehow pursuing the same goal in particle physics for years, although the amount of interactions with other users is quite limited at the present time (summer days, small SteemSTEM community, etc. Who knows).
Personally, it was a bit hard to read at the beginning because of the large usage of jargon. But at the end, the post is fairly understandable and I do not end with any particular question.
Yeah, I hope so too. Esp given the drive you started about up voting comments on the post, it should be helpful to motivate people.
Also, thanks for pointing out the use of jargon. I think I got a bit carried away with the paper and went ahead and presented it like I would do in our lab meet. But I have noted yours and @agmoore2 feedback. I will try to keep it easier to understand from the initial point to get non-biologists equally interested from beginning to end. Thanks once again.
Posted using Partiko Android
Voting comments will in principle bring no rewards with the next HF, unfortunately (if I have well understood it... I didn't invest long on that ;) ).
This post has been voted on by the SteemSTEM curation team and voting trail. It is elligible for support from @curie and @utopian-io.
If you appreciate the work we are doing, then consider supporting our witness stem.witness. Additional witness support to the curie witness and utopian-io witness would be appreciated as well.
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Thanks for having added @steemstem as a beneficiary to your post. This granted you a stronger support from SteemSTEM.
Thanks for having used the steemstem.io app. You got a stronger support!