BOTANICAL MICROTECHNIQUE // Morphoanatomical techniques and characterizations

In this new issue, I will socialize with the entire academic community of the social network Steem, the different techniques used in the preparation of plant micro-samples, intended for studies of anatomical and morphological characterization, in this opportunity we will address the main procedures used by optical microscopy at the laboratory level.
Introduction
Histological studies of the plant area represent a recurrent practice in botanical research, because they allow characterizing microscopic structures at tissue level, and relate morphological representation with the functionality of each histological group, hence, there are different specific procedures for the preparation of plant samples, as well as microscopic observation methods, which can determine the differentiation of plant tissues.
However, the set of procedures applied to characterize microscopic structures at plant tissue level is called Botanical Microtechnique, this area of botanical histology, aims to prepare and confer optimal conditions to plant micro-samples, in order to observe, examine and analyze in detail the morphological and anatomical components, through specific microscopy techniques.
In attention to these considerations, the objective of the present post pursues to socialize the main technical procedures, used to carry out morphoanatomical characterizations in vegetable micro-samples, complementing for it, determinations at laboratory level.
Plant Histology
The area of botany, which is in charge of the meticulous study of plant tissues, is called Plant Histology, a scientific discipline focused primarily on the structural and morphological identification of each set of biologically constituted cells, and thus be able to deduce, understand and analyze the functions of plant tissues resulting from these associations, as well as their relationship with each organographic structure.
Botanical Microtechnique
Within plant histology, the set of procedures applied to characterize the morphoanatomical structures of plant micro-samples is known as plant microtechniques, the purpose of which is to observe with specification and detail the plant tissues, determining the morphogenic changes since the appearance of the zygote, conformation of specialized organographic structures and the senescence of these.
The procedures applied to characterize the morphoanatomical structures of plant micro-samples, namely; histological cuts, histological coloration, preparation of plant micro-samples, histological assemblies and observation.
Histological Cuts
The histological cut is understood to be the thin fraction, or organographic part of a vegetable species that has been sectioned, with the objective of describing it anatomically and morphologically, detailing its cells and tissues, within the histological techniques used, the following are found; a) Mechanical cut (use of microtomes), b) Freehand cut.
Cuts with microtomes
Histological cuts made with microtomes are of great precision, basically because these modern devices manage to divide the vegetable micro-samples, in very thin slices, with structural precision and in sections that can oscillate between 1 and 50 microns, this according to the capacity of the instrument and research objective.

Fig. 2 Microtome. Public domain – credits. Author: Luigi Chiesa, 2006 / CC BY-SA 3.0
Freehand cuts
It is important to point out that within the teaching of histological botany, the cut most used in laboratory practices is precisely that of a raised hand. This conventional technique consists of holding the plant material between the thumb and index fingers of one hand, resting the knife, blade or scalpel in one of the corners of the index finger of the other hand, and keeping the arms fixed as close as possible to the body, it should be noted that the cuts are made in the direction of the body, and the number of cuts should be sufficient, this in order to select the thinnest.
Orientation of histological cuts
The orientation taken to perform the division of plant micro-samples largely serves the purpose of morphoanatomical characterization of the specimen subject to evaluation. However, the orientations of histological slices widely used within the observations, by optical microscopes, are as follows;
A. Cross section
This histological cut is made perpendicularly to an imaginary axis that crosses the sample, dividing the vegetable micro-sample into an anterior and a posterior section, also called the basal section.
B. Longitudinal cut
The orientation of the longitudinal cut is made parallel to the imaginary axis that crosses the vegetable micro sample, separating a dorsal region from the ventral.
C. Tangential cutting
This is not a histological cut per se, since the procedure really consists of removing a fraction tangentially to the axis of the vegetable micro-sample.

Fig. 3 Tangential cut in Eichhornia crassipes foliar sheets. Author: @lupafilotaxia.
Histological coloration
In plant microtechnology, the process by which plant micro-sample is dyed by a substance called dye, is known as histological coloring, it is necessary to indicate that there are different types of dyes, however the most used in botany are;

Fig. 4 Colorants used in the UNESUR Agricultural Botany Laboratory. Author: @lupafilotaxia.
1. Safranin
Safranin is a basic Colorant, widely used in plant histology, because it allows to dye lignified, cutinized, suberified and chitinized cellular structures, and also to color chromosomes and nuclei.
2. Floroglucine
This organic dye is used to dye lignified cell walls, so it is used in those tissues that present; xylem and sclerenchyma.
3. Lugol
Lugol, a compound obtained from the dissolution of molecular iodine and potassium iodide, is commonly used for the recognition of starch granules, a substance that react chemically with Lugol, taking a blue-purple coloration.
4. Sudan III
Sudan III dye is the most widely used compound in plant histology to colour cutin and suberin, mainly due to its ability to react with lipidic substances.
Preparation of vegetable micro-samples
There are different methods involved in the study of plant micro-samples, optical microscopy being one of the most widely used techniques in the detailed, structural and morphoanatomic observation of plant tissues, while the processes involved in the preparation of the material may be developed separately, or in a combined and histologically identified as follows:
A. Fixing
In plant histology, the procedure performed with the objective of stopping metabolic processes at the cellular level is known as fixation, or in other words, not so technical, simply killing cells as quickly as possible, simultaneously preventing possible morphological modifications, i.e., the ultimate goal of the procedure is to stabilize cellular components.
In the search to achieve stabilization of cellular components, generally used substances or mixtures of fixing substances, related to the specimen or quality of plant micro-samples (For example, vascular plants, mosses and some hepatic, can be fixed in 70% Alcohol, or in Formol - Alcohol - Acetic Acid), on the other hand, it is advisable to use two types of fixatives, this to achieve a good fixation, regardless of the size of the sample and its resistance to infiltration.
Types of fasteners
According to the components of the fixator can act two types;
1.- Acid, characterized by optimal preservation of chromatin, nucleoli and mitotic spindle.
2.- Basic, whose specificity preserves well the nuclei, the nucleoplasma, the mitochondria and the vacuoles.
B. Hardening
This procedure is carried out with the aim of giving vegetable micro-samples resistance, essentially to be able to section them, either through the use of knives, scalpels or microtomes, paraffin being one of the main substances used to harden the material, which will be observed with the optical microscope, while resin-based substances are used in those cases where observations are made using electronic microscopes.
C. Preservation
Preserving vegetable micro-samples implies the use of preservative substances such as 70% Alcohol or, failing that, Formol - Alcohol - Acetic Acid, in order to avoid the appearance of mould (filamentous structures of fungi) and rottenness of the plant material under study.
Histological assemblies
After developing all the procedures described above, we proceed to execute the assembly, a technique that allows us to preserve the structures for minutes, hours or even years. In this section, it is important to mention that the success of any assembly, before taking it to the optical microscope for observation and determination, is characterized by cleaning the object slide, depositing a drop of distilled water, placing the micro-sample of plants, covering it with an object cover.
The optimization of any assembly technique invariably depends on the type of preparations, whether temporary or permanent.
A. Temporary preparations
Temporary preparation is a very simple histological technique because, at the moment of its execution, it is not necessary to stop the metabolic processes of the vegetable micro-sample, that is to say, it is not necessary to use fixing, hardening or preserving substances.

Fig. 5 Temporary assembly of vegetable micro-samples of Limnocharis flava foliar laminae. Author: @lupafilotaxia.
B. Permanent preparations
Permanent preparations are used in those cases where morphoanatomical determinations can be studied at a later date, which is why it is necessary to previously fix, harden and preserve the vegetable micro-sample with special cutting and staining techniques.
Morpho-anatomical study
In order to exemplify part of the socialized content on plant microtechniques, structural and morpho-anatomical descriptions were developed using optical microscopy methods, using the UNESUR Botany laboratory.
Processing of vegetable micro-samples
For the purposes of this post, 03 (three) vegetable micro-samples, previously determined from leaf sheets of Eichhornia crassipes, Limnocharis flava, and adventitious roots of the species Tradescantia spathacea, were considered for the study. The selected procedure was: Temporary preparation, which consisted of placing a drop of distilled water on an object slide, placing the sample to be observed on the drop of distilled water, placing an object cover on the sample, and finally removing the excess water with absorbent paper.
Histological observation and orientation
Each histological segment considered came from previously unidentified plant micro-samples, based on transverse and tangential freehand sectioning, while the observations were made in OPTIKA brand optical microscopes, with the purpose of determining each fraction, using histological approaches first at 4x and then at 10x.
Morpho-anatomical results
Even though the histological sectionings were not performed with the help of the microtome, the cuts obtained by the freehand technique presented thin segments, complete and in acceptable ranges, that allowed to visualize and identify the associations of cells in each one of the considered vegetable micro-samples.
The cross section of the E. crassipes leaf blade showed (a) smooth epidermis of rectangular appearance, thin cell wall, without trichomes, (b) parenchymal cells forming rows, and longer than epidermal cells, (c) compact vascular beam underneath the fundamental tissue, (d) rounded, interlaced and forming rings that constitute (e) air chambers (Fig. 1) .
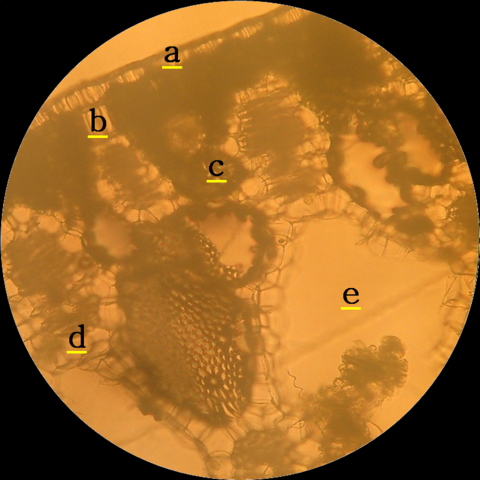
Fig. 6 Cross-section of E. crassipes foliar sheet. Author: @lupafilotaxia.
The morphoanatomical structure, determined in the tangential section of the foliar lamina of L. flava, showed (a) thin cell wall, (b) pore or open ostille, (c) occlusive cells in a turgid state, (d) fundamental cells slightly rounded and (e) accompanying cells of hexagonal geometry (Fig. 2).

Fig. 7 Tangential section of foliar sheet of L. flava. Author: @lupafilotaxia.
The morphology and anatomy, of adventitious roots in T. spathacea, showed (a) exodermic cells and (b) small and rounded endodermic cells, (c) grouped phloematic cells, (d) xylematic cells forming rings and (e) medullary parenchymal cells (Fig. 3) .

Fig. 8 Cross section, of adventitious roots in T. spathacea. Author: @lupafilotaxia.
SCIENTIFIC CONTRIBUTIONS OF THIS PUBLICATION
- The structural and morphoanatomical determinations, obtained from the histological sections considered, present diffuse characteristics that prevented the precise visualization of cell layers and the recording of morphometric variables. However, the topographic patterns observed in each histological segment evaluated allowed the identification of specific cell associations, a useful element for diagnostic and taxonomic interpretations; temporal preparation, transverse and tangential sectioning, freehand cuts and observation by optical microscopy, could be considered in the experimental checks prior to morphoanatomical characterizations with specific staining, specialized assembly and cutting with microtome.
BIBLIOGRAPHICAL REFERENCES CONSULTED AND CITED:
[1] Curtis J. Microtecnia Vegetal. 2 ed. Editorial Trillas. México, D. F.1986;106.
[2] Izco J., y Barreno E. Caracteres taxonómicos: Morfología y anatomía de órganos vegetativos. Editorial Edigrafos. Madrid. 2000.
[3] Johri B. Embryology of angiosperms. Springer-Verlag, Berlin. 1984.
[4] Lindorf H. Eco-anatomical wood features of species from a very dry tropical forest. IAWA Journal. 1994;15;4:361- 376.
[5] Roth I. Microtécnica Vegetal. Ediciones de la Biblioteca, U.C.V., Caracas. 1964.
ATTENTION / Readers and Followers
If you want to read more scientific articles of excellent academic quality, do not waste time, and visit the label #steemSTEM, is a large community that values and promotes scientific content, mainly in the areas of Science, Technology, Engineering and Mathematics.

INVITATION
Dear reader, if you wish to obtain more information about it, join our server in Discord
OBSERVATIONS
✔ The POST 📧📨 was uploaded using the official app of https://www.steemstem.io
OBSERVATIONS
✔ The POST 📧📨 was uploaded using the official app of https://www.steemstem.io
You can visit the official app of our community
https://www.steemstem.io

0
0
0.000
Excelente publicación! Saludos
@tipu curate
Upvoted 👌 (Mana: 0/20 - need recharge?)
Thank you for your support.
To listen to the audio version of this article click on the play image.

Brought to you by @tts. If you find it useful please consider upvoting this reply.
Hello,
Your post has been manually curated by a @stem.curate curator.
We are dedicated to supporting great content, like yours on the STEMGeeks tribe.
If you like what we are doing, please show your support as well by following our Steem Auto curation trail.
Please join us on discord.
Thank you team @stem.curate
Very good material Professor Luis as always we are used to all readers. I really enjoy reading your topics about botanicals.
There was a user here in steem who published very similar to you, but the content was about agriculture, especially about animals. I don't remember his name but both connected very well.
Esta publicación ha sido seleccionada para el reporte de Curación Diaria.
Have a nice day, @lupafilotaxia.Your post was selected to win some extra TLNT tokens, powered by The Talent Club.That means you got talent!Enjoy it!Enhorabuena, su "post" ha sido "up-voted" por @dsc-r2cornell, que es la "cuenta curating" de la Comunidad de la Discordia de @R2cornell.
This post has been voted on by the SteemSTEM curation team and voting trail. It is elligible for support from @curie and @minnowbooster.
If you appreciate the work we are doing, then consider supporting our witness @stem.witness. Additional witness support to the curie witness would be appreciated as well.
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Thanks for having used the steemstem.io app and included @steemstem in the list of beneficiaries of this post. This granted you a stronger support from SteemSTEM.