PARTICLE PHYSICS: Discovery of Particles and the Existence of Energy, Matter and Antimatter.
Scientists once thought that atoms were
indivisible – the word ‘atom’ means ‘cannot be cut’. They looked more closely and found electrons, protons and neutrons. Again, they thought these were the
smallest particles. Then physicists found they were not but were divisible
into a whole array of even smaller particles.
Where does this subdivision end, and will we ever know we have found truly fundamental particles? These are questions to which there is so far no definite answer.
The ideas in this article
In a study of the world of subatomic particles, we are entering a very strange world indeed. What we see about us in everyday life is on the whole, stable matter.
The theory of how the world of stable matter came about is called the standard model. This model says that about 15 billion years ago there was a ‘singularity’, a highly unstable point which was the entire Universe. Then, time and space began in the Big Bang. I’ve discussed in more detail with this stage in the development of the material of the Universe in one of my previous articles.
Today, researchers into the fundamental particles of matter use very large particle accelerators at international laboratories such as CERN (Centre Européen de Recherche Nucléaire) and other large international and national laboratories.
However, the most highly energetic particles come free: they are the cosmic rays that arrive from outer space – particles (most of thenm protons) which have been accelerated in the magnetic fields of exploding stars to speeds approaching the speed of light.
The practical application of particle physics is not as remote from everyday life as you might imagine. For instance, particle accelerators and detectors are routinely used in hospitals to diagnose and treat diseases. Archaeologists and art researchers do detection work with these expensive pieces of equipment. They have also been used to discover and improve new materials for industry and the home, such as superconducting materials, semiconductors and video screen surfaces
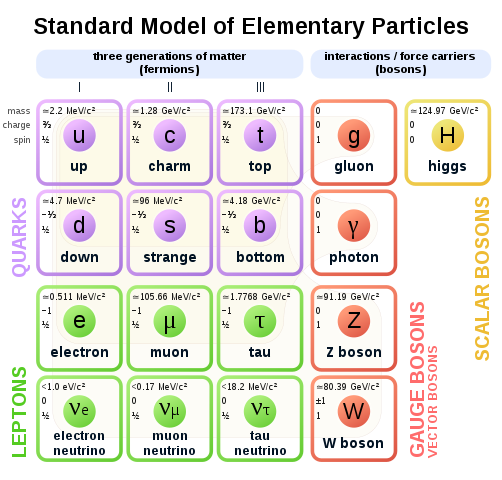
This article deals with how particles were discovered and are detected and manufactured in experiments with very large machines – the particle accelerators. It describes how physicists explain and predict the events of the subatomic world, which will involve quantum physics and relativity theory, the existence of both matter and antimatter, deep inelastic scattering and how events can be summarized in Feynman diagrams. You will learn about the types of particles that exist or are postulated to exist (hadrons, baryons, quarks, leptons, bosons) and the conservation rules that determine the reactions and relationships between them.

The particle content of the Standard Model of Physics. Cush, Public Domain
What you should know before tackling this article
You should know the following things about the size and nature of a typical atom:
- An atom is about 10-10 m across and consists of a small central nucleus just 10-14 m across surrounded by a cloud of wavy particles’, the electrons.
- The nucleus contains practically all the mass of an atom and (except for hydrogen) is made up of two types of nucleon: protons and neutrons. Protons are stable, but free neutrons (not in an atom) decay. This can also happen inside a nucleus which has too many neutrons.
- An electromagnetic force binds electrons to the nucleus.
- A strong nuclear force binds nucleons together inside the nucleus.
- There is also a rather more mysterious weak nuclear force that is (for example) involved in beta decay.
This post has many terms and ideas that may be unfamiliar to you. I’ll make it as comprehensible as possible. Take it step by step and check your understanding before moving on.
THE SIMPLE ATOM GETS COMPLICATED
The simple Rutherford-Bohr model of an atom had three particles: neutron, proton and electron. But the observed energy spectrum of beta particles emitted in radioactive decay showed the need for another particle, the neutrino, which carries off both energy and momentum. This is a very small, uncharged particle, with a mass of zero or close to zero.
Then the British theoretical physicist Paul Dirac said in 1930 that, mathematically, each charged particle had to have a matching antiparticle, of identical mass but of opposite charge. So there had to be an electron with a positive charge. This anti-electron or positron was first observed experimentally in 1932.
Again because theory suggested it, the Japanese physicist Hideki Yukawa proposed in 1935 that another particle ought to exist, with a mass between that of an electron and a nucleon. This was called a meson (the Greek word for ‘middleweight’). In fact, Yukawa and his colleagues found two ‘mesons’. One had the properties predicted by Yukawa and is now called a pion or pi meson. This is a genuine meson, as the name is now used. The other turned out to be a kind of giant electron now called a muon.
TOO MANY PARTICLES?
More and more ‘mesons’ – middleweight particles – were discovered in the next thirty years, together with other particles which were more massive than the nucleons. This was all very confusing, and rather like the state chemistry was in before the idea of a Periodic Table and the theory of electron shells. The equivalent theory in particle physics is the ‘standard model’, involving the particles discussed in this post.
The first new particles discovered were thought to be the building blocks of ordinary matter and were called fundamental particles. But by the 1960s, there were even more of these ‘fundamental particles’ than there were elements in the Periodic Table! Most existed only for extremely short periods of time. To understand and categorize them, physicists needed to identify their properties and compare them. We will look at this shortly, and describe what are now seen as fundamental particles. But first we look at how particles are discovered.
DISCOVERING PARTICLES
The electron was the first subatomic particle to be discovered, in 1397. The cathode ray tube technique used then is used in particle accelerators today. In the cyclotron, for example, charged particles are accelerated in a magnetic field at right angles to their direction of motion, which makes them move in the arc of a circle. In a given magnetic field, the radius of the circle depends on the charge, mass and speed of the particle.
These accelerated particles are then used in collisions to produce other subatomic particles, rather like the formation of particles by cosmic rays. Particle detectors enable the newly formed particles to be located and identified. It is not these particles that are seen, but their tracks formed as ionized particles of the material they pass through. As we shall see, neutral particles leave no tracks themselves, but other clues of their presence can be detected.
ENERGY AND MATTER
Accelerated (highly energetic) particles are useful for the investigation of new particles because of a consequence of the special theory of relativity that energy and mass are interconvertible.
The figure below shows a bubble chamber, a type of detector of charged particles important historically but now obsolete. An incoming proton travels at a speed very close to the speed of light. It has very high kinetic energy. As it collides with a stationary proton (a hydrogen nucleus), this energy creates all the new particles and provides them with the kinetic energy to move away. Following the Einstein relation E = mc2:
mass-energy of incoming proton → masses plus kinetic energies of new particles produced

In an example of such a collision (at A). The
bubble tracks of some particles arc downwards, and others arc upwards. The
small tightly curved spiral is from an electron knocked violently out of an
atom by the incoming proton, showing that particles curving upwards are
negatively charged, while those curving downwards are positively charged. At B,
two particles appear as if from nothing. They are a newly created proton and a
negative meson known as a pion, symbol π-1.
We have to guess that these two particles were formed at B by a decay or a
collision involving a neutral particle that left no track as it travelled from
the first collision at A to the start of the two new tracks at B.
SOME COLLISIONS AND DECAYS
Particles that are observed in a detector, such as a bubble or cloud chamber tend to do one of two things:
- In simple collisions, they collide with a nucleus already there, and then may possibly break up the nucleus and/or create new particles.
- They self-destruct by ‘decaying’ into other particles or by colliding with an antiparticle.
I will now look at both kinds of events in more detail.
Simple collisions
Look at the diagram below. It's showing the structure of a cloud chamber, where collisions between alpha particles and the nuclei of hydrogen takes place. Helium and nitrogen as observed in the cloud chamber. The tracks in this and similar diagrams have been (redrawn and) artificially coloured to make things clearer.
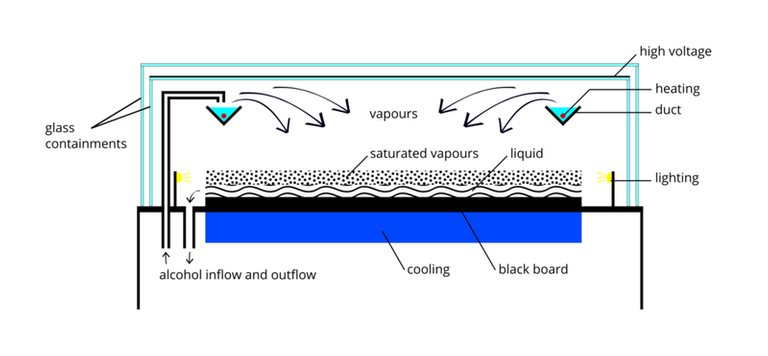
The pictures show that the angle between the tracks of the alpha particle and the nucleus after collision is decided simply by the ratio of the mass of the alpha particle to that of the nucleus it hits.
For another simple collision that indicates different masses, in which a heavy cosmic ray particle, in this case a sulphur nucleus (red track), collides with a nucleus in the photographic emulsion. The heavy blue tracks are nuclei, the thin yellow ones are the tracks of smaller, less ionising particles identified as pions.
Particles that decay
Unstable particles are like unstable (radioactive) nuclei: for a particular particle we can predict what will happen, but not when. The newly created particles will decay, eventually, into the stable particles of matter, which are protons, electrons and neutrinos. Occasionally, the electromagnetic particle the photon is emitted, or the decay is completely to photons, leaving no matter behind. As with radioactive nuclei, we can be certain that half the number of any type of unstable particle will decay in a particular time, so we use the idea of the half-life of the particle.
We now know that every known particle has its antiparticle, equal and opposite in its properties. For example, the positron is an antiparticle, exactly the same as an electron but opposite in charge. Particles and antiparticles make up what we call matter and antimatter.
When an antiparticle collides with its ordinary-matter particle twin, they annihilate each other. Their mass-energies are converted to new particles, namely photons and massive particles, that appear in the detector.
MATTER AND ANTIMATTER
When a particle meets its antiparticle there is instant annihilation of mass. The mass-energy of the pair appears as new particles (supplying both mass and kinetic energy) and/or radiation. The most commonly observed matter-antimatter collision is that for electrons:
electron + positron → 2 photons
e- + e+ → 2γ
Why two photons? This is because photons carry not only energy but momentum, as explained below. Momentum has to be conserved. If we think of the two particles meeting head on with the same speed, the original momentum adds to zero. If the result was a single photon, the momentum would not be zero. The momentum can only be the same before and after the collision with two photons carrying equal quantities of momentum travelling in opposite directions.
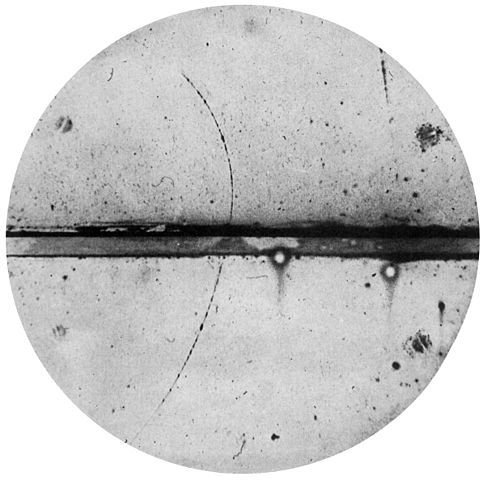
Cloud chamber photograph of the first positron ever observed. Carl D. Anderson, Public Domain
Pair production
Just as an electron-positron pair can annihilate to produce photons, so photons with enough energy can ‘decay’ into these and even more massive particles. The figure below shows such an event seen in a bubble chamber; it is called pair production. The event occurs only in a strong electric field – near a charged nucleus for example.
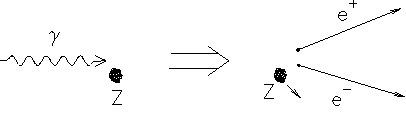
Watch out for the conclusion in my next post. Till then, I remain my humble self, @emperorhassy.
Thanks for coming.
REFERENCES
https://www.britannica.com/science/particle-physics
https://www.nature.com/subjects/particle-physics
https://www.sciencedaily.com/terms/particle_physics.htm
https://en.wikipedia.org/wiki/Particle_physics
http://content.time.com/time/health/article/0,8599,1990758,00.html
https://www.nap.edu/read/6045/chapter/3
https://simple.wikipedia.org/wiki/Atom
https://en.wikipedia.org/wiki/Timeline_of_particle_discoveries
https://en.wikipedia.org/wiki/Cloud_chamber
https://www.infoplease.com/encyclopedia/science/physics/concepts/cloud-chamber
https://www.britannica.com/science/bubble-chamber
https://en.wikipedia.org/wiki/Bubble_chamber
https://home.cern/science/physics/antimatter
https://en.wikipedia.org/wiki/Antimatter
https://www.britannica.com/science/pair-production
https://en.wikipedia.org/wiki/Pair_production
Hello,
Your post has been manually curated by a @stem.curate curator.
We are dedicated to supporting great content, like yours on the STEMGeeks tribe.
If you like what we are doing, please show your support as well by following our Steem Auto curation trail.
Please join us on discord.
This post has been voted on by the SteemSTEM curation team and voting trail. It is elligible for support from @curie and @minnowbooster.
If you appreciate the work we are doing, then consider supporting our witness @stem.witness. Additional witness support to the curie witness would be appreciated as well.
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Thanks for having used the steemstem.io app and included @steemstem in the list of beneficiaries of this post. This granted you a stronger support from SteemSTEM.
Congratulations @emperorhassy! You have completed the following achievement on the Steem blockchain and have been rewarded with new badge(s) :
You can view your badges on your Steem Board and compare to others on the Steem Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOP