Trouble Brewing in the Lab
Well, this was an unexpected surprise from my department supervisor.
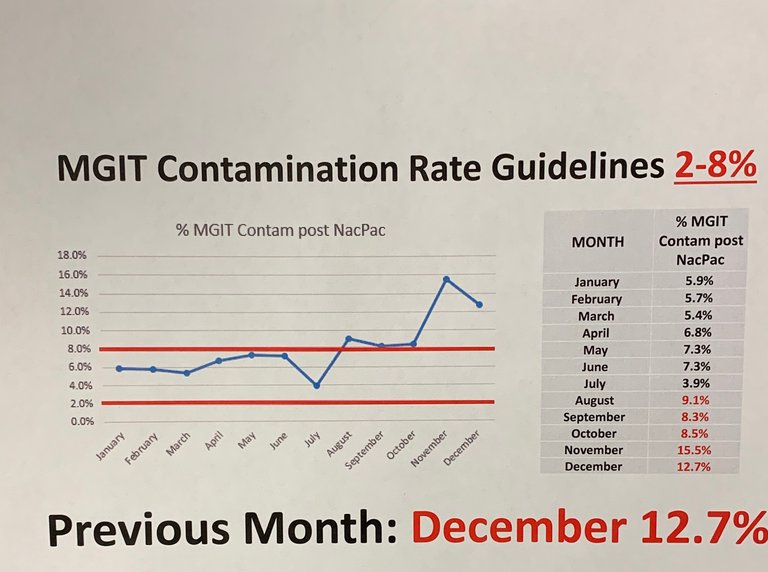
Without knowing anything, one could tell something terrible had happened. Our quality control is outside of accepted range. Internal investigations took place to figure out what went wrong for months.
The percentages you see here relates to a protocol for isolating acid fast bacilli (AFB). This class of bacteria includes those that cause tuberculosis and leprosy. This post won't discuss the workflow in details, but decontamination is a key step in the process. This is due to their slow growing nature compared to other microbes.
The protocol is also used in isolating deadly organisms such as Nocardia species.
The problem
My supervisor isolated the issue. The problem came from the decontamination reagent. At my lab, we use NAC-PAC Red from AlphaTec to process our non-sterile samples.

Another product photo you see.
As you can see from the image, the product includes a vial of pellets next to the bottle. The pellets are vital part of the solution. We have found a surplus of them in our storage. This suggested that some staff members were not preparing the reagents as instructed. This meant that many patient samples winded up having microbes we don't want growing.
What does this mean for the lab?
This means the lab is spending more time decontaminating samples than expected. We were wasting time and resources per test. This increases the number of false positive cultures in our work flow.
I sounded like a corporate shill in that paragraph, but things aren't cheap. And it sucks when you have to spend more resources when you didn't have to.
To remedy the blunder, every staff in the department will receive retraining. In addition, every NAC-PAC bottle will have its corresponding pellets taped to it from now on.
I haven't heard what the medical director has to say in this matter. I know my supervisor has increased the frequency of reporting those statistics. He is definitely going to compile those specific stats more than once a year going forward.
What does this mean for the patients?
Having to do extra decontamination steps affects turn around time. This means we cannot eliminate the possibility of AFBs as soon as we'd like. While cultures for AFB can last up to six weeks, this folly increases the average turn around time.
On the grand scheme of things, we are within the accepted time frame. But, this means the patients would have to wait longer before receiving treatments. In health care, the philosophy is to reduce the patients' suffering, not prolonging it.
The most important thing is, the mistake did not cost the accuracy of our results. It was definitely annoying to do more work than needed.
What does this mean for physicians?
They may not know any better. In my previous posts, I have mentioned that clinicians don't often know the lab processes. But, I would imagine it is frustrating for doctors to wait for results on an ill patient.
The only physician I'm worried about is our medical director. It's gonna be fun hearing from him. We have an early scheduled department meeting coming up.
Bonus
On the chart, you may have noticed that the accepted range is 2-8%. You might be wondering what happens when you go below that mark? The short answer is that it's also bad because that means we have false negative cultures. False negatives mean we over-decontaminated patient samples and killed all the microbes.
In my limited understanding, false negatives are much worse in this scenario. A lot of the AFB infections have long-term consequences if left alone. I want to mention that I don't know what precautions clinicians follow to mitigate that sort of mistake on the lab's end.
To listen to the audio version of this article click on the play image.

Brought to you by @tts. If you find it useful please consider upvoting this reply.
Sounds like that was the problem from the get go.
Sounds like another problem from the get go.
Sounds like another problem from the get go
Sounds like some people shouldn't have been hired in the first place.
If it wasn't for the fact people could die over shit like this I'd be saying thank god this isn't a criminal forensic's lab.
Sounds like someone needs to reread and understand things before acting like a know-it-all again.
!dramatoken. 0.o
What frickin' more does anybody need to read? It's was YOUR words. According to that first paragraph and everything I highlighted before some people in the lab were inept at doing their jobs. I am getting quite tired of you flagging my comments. This is why people don't engage. You wrote something and I gave a opinion that was spot on in relations to what you wrote. If you don't remove the flag I am not going to follow as you are a waste of my time.
Very very interesting, thanks for posting this.
A friend of mine had a biopsy done and was booked in for a double mastectomy.
Just before they performed the surgery, they found a problem at the lab. Did more biopsies and was found to be clear. Months after that there was a news story saying there was more than 400 women misdiagnosed.
Your article made a little clearer how such events can occur.
Given your work it seems strange and a shame that you choose to support the misanthropic element here.
Or is there is a misanthropic element to your work there as well?
I don't work in histology, so I can't tell you the exact process that happens there.
In this particular case, no misdiagnosis happened. It simply increased the average turn around time. When we decontaminate samples, the timer for incubation needs to restart. Non-AFBs can grow pretty fast, they can be detected within 1-2 days.
We still reported results within the acceptable time frame as outlined.
Your comment serves other motive. I can only answer what I know.
Ah yea i have no idea about the biopsy process. From your article I assumed that some kind of reagent would be used, but from your reply maybe not?
You must at least know why you align yourself with the misanthropic element here!
What is it exactly that you imagine you are protecting?
lol
Not entirely sure why I like that so much.
But maybe its because its Not Secret Anymore : )
It's more complex than you think.
But your version is indeed more interesting.
I am absolutely convinced that it is complex and far more interesting than anything I could possibly dream up. Having said that, I am quite sure it is not so complex that i would not be able to comprehend the intricacies and numerous subtleties.
Maybe you feel incapable of articulating the situation clearly enough for others to understand? Don't concern yourself with that thinking, I will get the gist of it just fine. But you knew that already
I love this low-tech, common sense solution. (Pin the mittens to the coat and they'll never get lost again).
An interesting, enlightening look into lab "culture":)
It's the easiest workaround.
Congratulations @enforcer48! You have completed the following achievement on the Steem blockchain and have been rewarded with new badge(s) :
You can view your badges on your Steem Board and compare to others on the Steem Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPVote for @Steemitboard as a witness to get one more award and increased upvotes!
Have a witness !BEER
View or trade
BEER.Hey @enforcer48, here is a little bit of
BEERfrom @isnochys for you. Enjoy it!Learn how to earn FREE BEER each day by staking.
This post has been voted on by the SteemSTEM curation team and voting trail. It is elligible for support from @minnowbooster.
If you appreciate the work we are doing, then consider supporting our witness @stem.witness!
For additional information please join us on the SteemSTEM discord and to get to know the rest of the community!
Please consider using the steemstem.io app and/or including @steemstem in the list of beneficiaries of this post. This could yield a stronger support from SteemSTEM.