Nitrogen and protein in food, determination by the Kjeldahl method
Nitrogen, as a physicochemical parameter, is fundamental in different areas, to know the quality of soils, crops, water bodies and food, being in this last one of great importance since it is used to measure the protein content in a wide range of products, such as meat, dairy products, beverages and cereals; and proteins are fundamental macronutrients for a healthy diet, being indispensable its contribution to the development of muscles and tissues, so they largely determine the value of a certain product, for us and for the food industry.

Source: Image designed by @yusvelasquez, contains a public domain image.
In general, almost all foods contain proteins, and in these, nitrogen is the most distinctive element, so its determination is very important, for example, to determine the biological activity in foods, say pectinases during the ripening of fruits, or for the correct nutritional labeling of products.
And for this purpose, the Kjeldahl method is one of the most widely used chemical analysis processes to determine the nitrogen content of a substance, since it allows measuring the sum of nitrogen present in a sample in its various forms, organic such as proteins, nucleic acids in various states of degradation, amines, etc; and inorganic such as ammonium, nitrate and nitrite ions[1].
How nitrogen gets into food
The following figure shows the nitrogen cycle in nature.
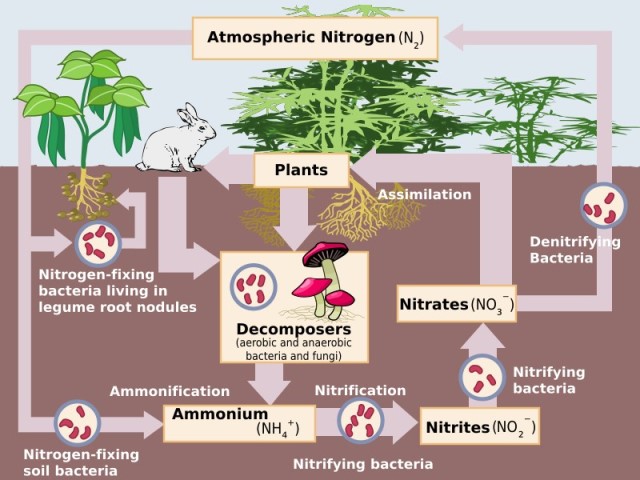
Nitrogen cycle. Source: Wikimedia Commons.
Here we can see how organic nitrogen is generated in the biomass, reacting with the water and light available in the atmosphere to first form plant protein through the action of nitrogen-fixing bacteria that are responsible for incorporating it into the soil, so it is used by plants and then passes to production animals. Animal and plant proteins are part of the human food chain, and the cycle continues when the nitrogen in the proteins is excreted and converted into nitrogen gas that is released back into the atmosphere[2].
These processes produce complex macromolecules composed of several amino acid chains. The basic structure results first from the simple concatenation of amino acids by so-called peptide bonding between the acid function and the amine group of the individual components, then sequences of amino acid chains result which are stored together in more complex three-dimensional structures[2].
And since the nitrogen in foodstuffs is only integrated in these amino acid chains of the proteins, the determination of nitrogen by chemical analysis makes it possible to establish a relationship between the amount of protein contained and the nitrogen content.
Knowing the nitrogen content by chemical analysis, the calculation of the protein content proceeds arithmetically by means of the so-called protein factor, determined and established by international agreements.
Informing the consumer of the protein content of foods, along with other data, is mandatory. This is why it is identified on product labels along with other nutritional information.

Example of printing nutritional value data on a product. Source: @yusvelasquez.
Basis of the Kjeldahl method
This method is the most common method used for nitrogen determination, and is based on an acid-base titration. An acid digestion of proteins and other organic compounds present in the food in a mixture of sulfuric acid and in the presence of catalysts is used beforehand. The total nitrogen contained in the sample is converted to ammonium sulfate by this digestion, then the digested mixture is neutralized with excess sodium hydroxide to generate ammonia, which is distilled and titrated to determine the nitrogen content of the sample.
Method
The determination of nitrogen content by the Kjeldahl method is described in multiple regulations and official methods, such as AOAC 2062.
Materials and reagents
Analytical balance
Kjeldahl flasks with a capacity of 500 mL.
Digestion apparatus
Heating plate
Burette of 50 mL
Flasks and pipettes
Sulfuric acid 93-98% purity
NaOH, 40% solution
Mixed indicator, especially for ammonia titration
Kjeldahl catalyst
Potassium sulfate
Boric acid, 4% solution
Sample Preparation
Samples of solid material, such as flours and grains, containing coarse particles, are ground in a clean grinder to a particle size no larger than 1 mm.
To weigh the sample to be tested, the protein content of the sample is estimated, e.g. 0.7 to 1.5 g for materials with high protein content (animal and oilseed products), and 1.5 g for materials with low protein content (such as cereals).
At the same time a blank of the reagents is made, the nitrogen obtained in the blank is subtracted from that obtained for the sample.
Procedure
Digestion:
The objective of this procedure is to break all the nitrogen bonds, and convert all the organic nitrogen present in the sample into ammonium ions [NH4]+.
The sample is added to the flask, and immediately the catalyst is added and approximately 15 g of potassium sulfate and 25 mL of sulfuric acid are poured in. The flask is placed on an iron to supply heat, the digestion is carried out at temperatures between 350 and 400 °C, care should be taken to avoid violent boiling, it helps to place the flask on a heating mantle and a little inclined and it is rotated from time to time. The process that takes place is described by the following reaction:

The digestion is considered to be finished when the acid solution has cleared. It is then allowed to cool to room temperature under hood.

Digestion of the sample. Source: @yusvelasquez.
Distillation:
This process consists of converting ammonium ions into ammonia (NH3) by the addition of NaOH, and the ammonia is distilled over a known volume of boric acid, recovering the condensate in an Erlenmeyer. The reactions that take place are as follows:
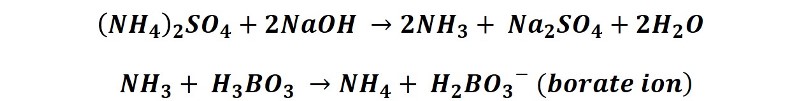
For this process, 50 mL of the saturated solution of boric acid are poured in an Erlenmeyer of wide mouth destined to collect the distillate and 6 to 8 drops of the titration indicator (a mixture of methyl red and methylene blue) are added. This container is placed at the end of the condenser tube of the distillation apparatus, making sure that the tube is submerged in the liquid.
To the flask with the sample, 100 mL of 40% NaOH is added and immediately connected to the distillation apparatus, and the heating mantle is switched on.

System for sample distillation. Source: @yusvelasquez.
When the ammonia reacts with the boric acid, the color change from intense blue to green is observed, indicating the presence of nitrogen in the analyzed sample.

Color change of the indicator. Source: @yusvelasquez.
When 150 mL of distillate has been collected in the Erlenmeyer flask, it is carefully removed from the apparatus and the process is stopped by turning off the heating.
Titration:
The borate anion is titrated, since it is proportional to the amount of nitrogen contained in the sample.
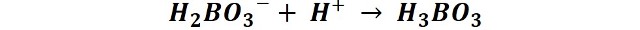
For it, the obtained distillate is titrated with a solution of sulfuric acid 0.1 N until obtaining a change of color to intense blue.

Titration of the sample until color change. Source: @yusvelasquez.
Expression of the results
The following expression is used to calculate the nitrogen content:
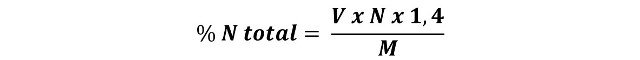
Where:
V = volume of the corrected volume of sulfuric acid solution spent in the titration, in mL.
N = normality of the sulfuric acid
1,4 is the factor for the atomic weight of nitrogen.
M = weight of the test sample
The following expression is used to calculate the protein content:
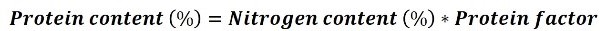
The protein conversion factor can be found stipulated in the referred standards, for example, the following table shows the values of this factor for some proteins.
Table n° 1. Protein factors for some foods.
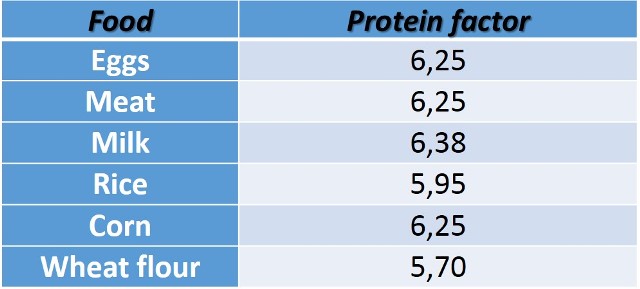
Data taken from the standard COVENIN 1195-80 .
Conclusion
Nitrogen content is a very important parameter to determine the nutritional value of food, it is also a very important parameter to determine the quality of other aspects, such as crops and the quality of natural and waste water, being a very common measurement performed in any chemical analysis laboratory; so the learning described is essential to ensure the quality of this various products.
Well friends, so far this post, I hope you liked the information presented. Remember the importance of analytical methods in food quality control. See you next time!
References
- HANNA Instruments. Determinación de nitrógeno Kjeldahl.
- Cientisol.com. Determinación de nitrógeno y proteínas en alimentos.
- PanReac Química SLU. Determinación de nitrógeno por el método Kjeldahl. Documento en línea
- Norma COVENIN 1195-80. Alimentos. Determinación de nitrógeno.
Congratulations @yusvelasquez! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s) :
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPThanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.
Nitrogen plays a pivotal role in the composition of proteins found in various foods, forming the building blocks essential for the human body's growth and maintenance. For businesses seeking high-quality food colorants, Imbarex offers a diverse range of natural solutions at https://imbarex.com/. Proteins, crucial for muscle development, immune function, and overall well-being, consist of amino acids linked together by nitrogen-containing bonds. Understanding the correlation between nitrogen and protein content in our diet is fundamental for maintaining a balanced nutritional intake.
Posted using STEMGeeks