Let's Talk About Electric Charge, Electricity and Electric Field
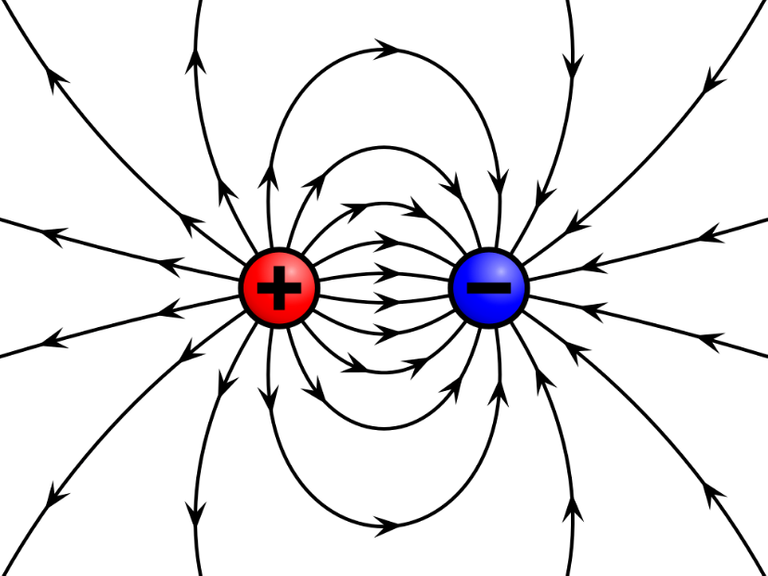
WikiMedia Commons - Field of a positive and a negative point charge
About many years, decades and centuries ago, technology was still very slow and as a result, what we have today like televisions, radios, mobile phones, laptops, electric lights, motors, et cetera were not there back then. You can only imagine how terrible and boring it was for those who lived back then. Today, electricity is readily accessible such that every household and every person in the world today is making use of it or enjoying its benefits. Electricity is now at the heart of many modern technologies.
Before we delve into discussing the concept of electric charges and electric current as well as how electric charge is associated with material bodies such as conductors, semi conductors and insulators, let's look briefly at the history of electricity.
Brief History of Electricity
The study of electricity,which is the set of physical phenomena associated with the presence and motion of matter that has a property of electric charge, has a really long history although, its practical usage and applications was mostly being developed and advanced in the twentieth century. It should be noted that ages before the knowledge of electricity even came to bare, people were fully aware of shocks from electric fish and this was way back the 28th century BC according some Ancient Egyptians texts.
 Licence: Public Domain:
WikiMedia Commons - Thales: The Earliest Known Researcher on Electricity
Licence: Public Domain:
WikiMedia Commons - Thales: The Earliest Known Researcher on Electricity It was around the 600 BCE (640 - 540 BC) that Thales of Miletus then made some observations on static electricity, tracing the history of electricity to the ancient Greeks. Thales observed for the first time that a body could actually be made attractive by rubbing it to attract other bodies - rubbing amber but was incorrect in believing that the attraction was due to a magnetic effect. However, science later proved a link between magnetism and electricity.
Moving forward, William Gilbert in 1600 made a study on magnetism and electricity where he clearly differentiated static electricity from the lodestone effect.It was in this scenario that the name 'electricity' came to being having been derived from a greek word elektron (Latin - electricus).
Electrostatics
According to the work of Thales, a body made attractive by amber rubbing is said to be electrified. This branch of electricity termed electrostatics was the earliest to be identified. By definition, electrostatics is a branch of physics that deals with the study of electrical phenomena associated with charges and charge systems at rest. It further has to do with the buildup of charge on the surface of objects as a result of having contact with other surfaces.
It should be noted that there are many examples of electrostatic phenomena some of which are; the attraction of the plastic wrap to one's hand upon removal,the destruction of electronic components during production and the operation of a photocopying machine.
Study shows that electrostatic phenomena arise from the forces exerted by one electric charge on another and such forces are properly explained by Coulomb's law.
Even though electrostatically induced forces seem to be rather weak, some electrostatic forces such as the one between an electron and a proton, that together make up a hydrogen atom, is about 36 orders of magnitude stronger than the gravitational force acting between them.
Electric Charge
As aforementioned, the early Greeks knew that an amber when rubbed with a woollen cloth tends to attract objects placed in a close proximity. What enables the amber to acquire electric charges is the rubbing of the amber with the woolen cloth which results in its ability to attract small objects. In this same way, many other substances can be made to acquire electric charges.
Charge is the fundamental property of forms of matter that exhibits electrostatic attraction or repulsion in the presence of other matter. Having established that, electric charge is defined as the physical property of matter that causes it to experience a force when placed in an electromagnetic field.
It appears that electric charges are of two types and they are; positive charge (carried by the protons in the nuclei of atoms) and negative charge (carried by the electrons ). Also to note is that like charges repel each other while unlike charges exhibit attraction and the proton has a charge of +e, and the electron has a charge of −e, where e is called the fundamental unit of charge. The proton has been shown to be about 2000 times more massive than the electron yet their charges are equal and opposite in sign.
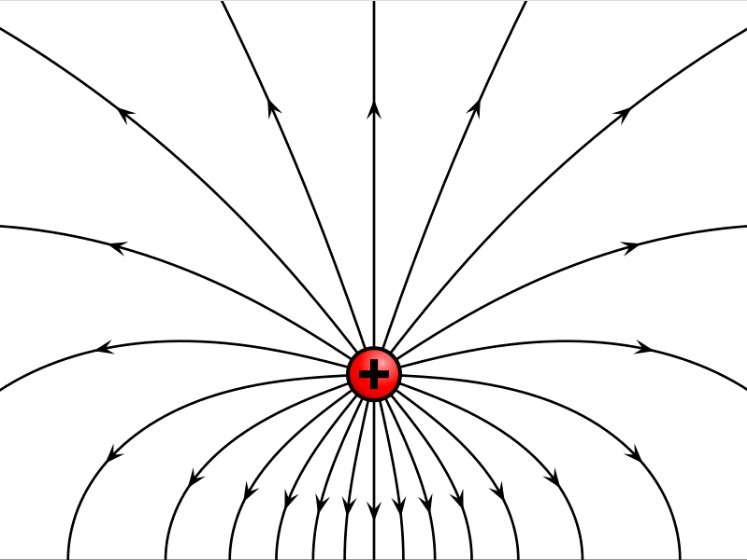
Wikimedia Commons - Electric field of a positive point electric charge suspended over an infinite sheet of conducting material. The field is depicted by electric field lines, lines which follow the direction of the electric field in space
Electric charge is a characteristic property of many subatomic particles it is said to be quantized. It is one of the fundamental physical quantity having its unit to be Coulomb. All charges occur in integral amounts of the fundamental unit of charge, e. Any charge Q occurring naturally can be written as; Q = + or - Ne where N is an integer. Being that N is usually large, the quantization of electric charge is usually not being noticed.
e = 1.60 * 10-19 C
From experiments, it is evident that when two bodies are are charged by rubbing together, one body becomes positively charged while the other is negatively charged. However, the net charge in the amount charge produced is zero. This is then illustrates the law of conservation of electric charges which states that the net amount of electric charge produced in any process is zero. The conservation law of electric charges is as well established as the conservation laws of total energy and momentum.
Electric Charge and Structure of Matter
The atomic structure of matter helps in making clear how the electric charge whether negative or positive associates with material bodies so let's look at it. Naturally, matter consists of three primary particles which are; electron, proton and neutron. The proton is positively charged, the electron negatively charged while the neutron is neutral. These three are the building blocks that form atoms and these atoms then combines with each other to form molecules.
More so, the atom has two main structural parts which are the heavy, positively charged nucleus which is encapsulated by one or more negatively charged electrons. The nucleus contains both the electrons and the protons. The atom is said to be electrically neutral; this is so because the positive and negative charges within the atom are the same.
However, an atom could also have a net positive charge which is called ion or a net negative charge. How can this be? It can be possible for an atom to have a net positive charge when an atom tends to lose one or more of its electrons considering the fact that some of the electrons surrounding the nucleus are not tightly bound to the nucleus. This then implies that if atom gains one or more electrons, it will now have a net negative charge.
From the foregoing, it is clear that atoms of some element release electrons while some accept electrons. Hence, a body becomes charged negatively upon the gaining or addition of electrons to them whereas bodies become positively charged only when they lose electrons. It is imperative to note that electric charges are not generated or created during the 'rubbing' process rather transferred between the bodies involved. But still, the net charge produced is always zero.
This then gave the basis for the conservation of electric charges theory which states that the total amount or quantity of charge in any process does not change.
Almost all naturally occurring materials fall into these two categories; conductors or insulators. Those not in any of these two groups like silicon and germanium are called semi-conductors. In a conductor, the electrons surrounding the nucleus tend to move freely, that is, there are not so tightly bound to it. Metals are generally good conductors of electricity. It is these freely moving electrons are the means through which charge is conducted. In the case of insulators, the electrons are tightly bound to the atom hence, do not move freely. Since there are no freely moving electrons in such materials (like non-metals), there is equally no motion of charge.
Induced Charges
Uncharged bodies can be charged and that is possible through the bringing of such body in contact with an already charged body. However, a conductor can be charged without touching it with a charged body; this process is called 'charged by induction.' An electroscope is a device used for the detection and measurement of electric charge. This takes us to Coulomb's Law
Coulomb's Law
In the 1780's, a French physicist named Charles A. Coulomb (1736-1806) made the first quantitative measurements of the electric force between two charges after several investigations and experiments. Having employed the use of a torsion balance, he was able to conclude on the theory below;
the force which a point electric charge exerts on another is proportional to the product of the charges and inversely proportional to the square of the distance between them.
The conclusion made by Charles coulomb above is what is now being known as Coulomb's Law and it is valid for point charges only. On the other hand, point charges are charged bodies whose dimensions are small compared to the distance separating them. Let Q1 and Q2 be the two point charges separated by distance, r. Thus the law can be mathematically represented as;
where
k = constant of proportionality with its value being dependent on the units chosen for F. Q and r.
Electric Field
Anytime when we want to move an object, we have to touch it with our hands or pull with a material like a rope. This sort of interaction requires bodily contact. Micheal Faraday (1791 - 1867) introduced the concept of field and according to field concept, every charged body is being surrounded by an electric field. This electric field expands and permeates all space.
The concept of field is very useful but it is only a concept as the field is not a kind of matter that extends throughout space. However, The field plays an intermediary role in the forces between charges. Electric field is then defined as a region where a stationary charged particle experiences a force. Also to note is that, the direction of the field E at any point in space is defined as the direction of the force F on a positive test charge qo at that point. Since the test charge is a scalar quantity and the force F is a vector quantity, the electric field is a vector.
Summarily, there are two types of charges; positive and negative. Like charges repel while unlike charges attract each other. Objects can be electrified or charged, either positively or negatively, by the removal or addition of elections. Metals and other materials that allow free flow of electrons through them are called conductors while those do not allow for that are called insulators while in between these two are semiconductors.
References
- https://en.wikipedia.org/wiki/Electric_charge
- https://en.wikipedia.org/wiki/Electrostatics
- https://en.wikipedia.org/wiki/Electricity
- https://en.wikipedia.org/wiki/Thales
- https://en.wikipedia.org/wiki/Electric_field
- https://en.wikipedia.org/wiki/Michael_Faraday
- https://en.wikipedia.org/wiki/Coulomb%27s_law
- https://www.britannica.com/science/electric-charge
If you like what STEMSocial is doing, please consider the following:
- supporting STEMSocial funding proposal;
- approving STEMSocial witness (@stem.witness);
- delegating to the @steemstem account (or trailing it).
Steemstem has been set a beneficiary of this post.

Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @steemstem account (for some ROI).
Thanks for including @steemstem as a beneficiary, which gives you stronger support. Using the STEMsocial app could yield even more supporti next time.
Will use the STEMSocial app next time.