The great internal war with the viruses - Episode 1 - The initial response to infection
I am the kind of person who doesn't like unknowns. But, I don't hate them, I prefer to explore them, and then make my choices based on those answers. But the question is how do you make your choices when you don't have time to explore. And I think this is the question that has taunted every one of us since the Covid19 pandemic, including the governments trying to make the policies. Every day, new information was pouring in. And I think that created even more confusion. Honestly, we are still learning a lot about this virus. But, I think we have also learned a lot of whatever was possible in this limited time.
I see that some of the questions that are raised regarding vaccines. Is natural infection better? How do we know these new vaccines work? What immune response is generated by vaccine vs natural infection? Should I get vaccinated if I already have previous exposure to Covid19?
Well, the answer to these questions lies in understanding what we know about viruses and the immune system in general. How do they interact? What does the immune system do? How does the virus respond and fool the immune system? What do we know about the response of the SARS-CoV-2 virus in particular? What inferences can we draw? With this in mind, I decided to create this series of posts dealing with the fight between the immune system and viruses. Once we have discussed some basics we will try to address those looming questions.
This is how it all starts
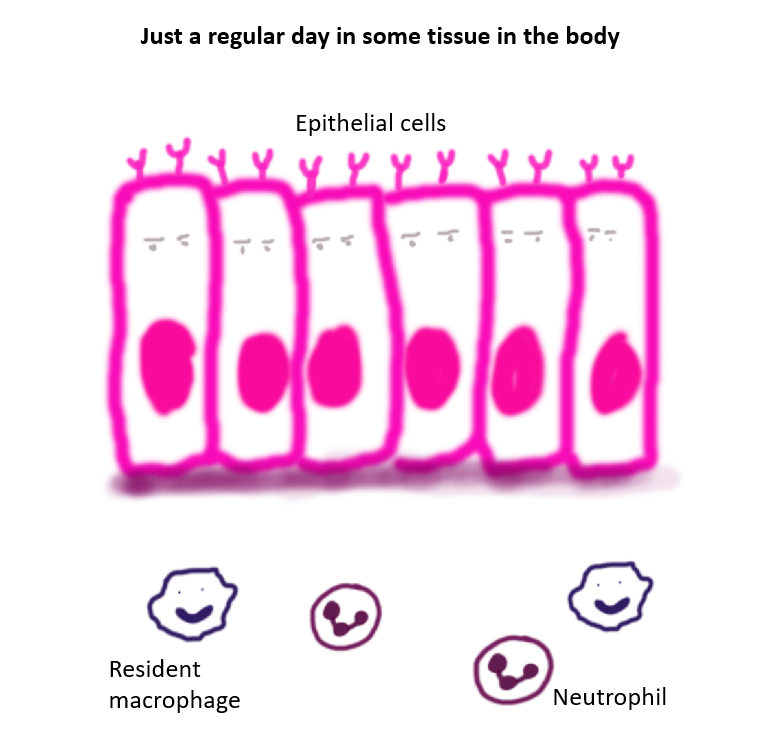
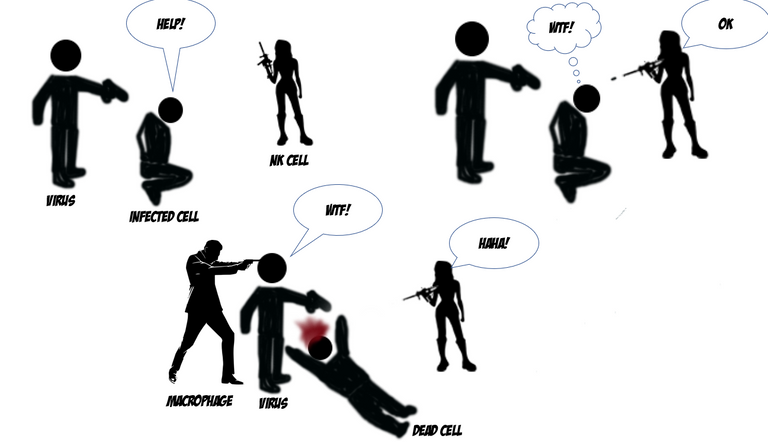
Immune system as a very strict team of law enforcement officers with specified tasks. Their main goal is to maintain peace within the society of cells that makes your body and protect it from illegal outsiders.
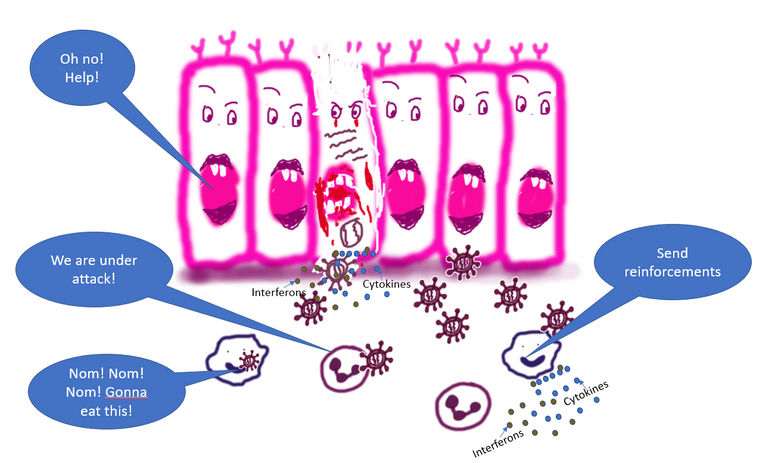
The virus attacks and starts infecting the cells. The infected cell start crying for help by secreting cytokines and interferons which warn the nearby cells and the immune cells
The innate immune cells recognize the pathogen and secrete more cytokines to call for reinforcements.
All above images are illustrated by @scienceblocks
The response from the infected cells.
When the virus infects the cell, the infected cell can detect the presence of the virus on its surface and inside 1. Accumulation of free viral genomic material in the cell cytoplasm is another signal for the infected cell to call for help. The overload of protein production machinery inside the cells also raise red flags. In response to the infection and stress induced by the virus replication, the infected cells secrete molecules called cytokines that warn all nearby cells and immune cells of the possible presence of the virus (such as TNFa, IL6, IL1b, CCL5, etc).
These cytokines include molecules that activate the nearby immune cells, molecules that travel to the bloodstream and lymph to call for more immune cells. The cytokines also include molecules called interferons, which are proteins whose major role is to fight viral infection 2.
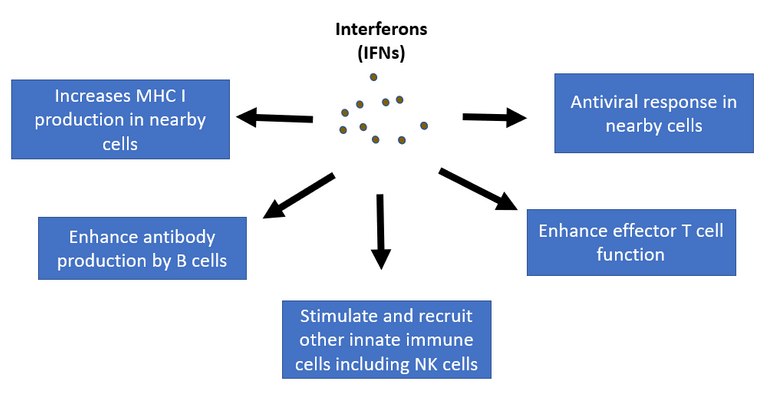
Interferons are like warning signals for nearby non-infected cells. In response to interferons, the cells turn up the gene expression of proteins involved in antiviral defence 3. They do so by making proteins inside the cells that block viral replication, increasing the production of inflammatory cytokines in nearby and immune cells hence amplifying the inflammatory signals 4. Apart from this they also make enhance the antibody production by B cells 5 and enhance the T cell function 6. On the surface of all nearby non-infected cells production of major histocompatibility complex (MHC) molecules 7. MHC are surface molecules that display pieces of various proteins being digested inside the cells, including viral protein. This is where T cells bind to and check if cells contain any foreign protein. (we will come to these soon).
It's almost as if what you would do when an invader gets into your house. You will call 911. You will scream to make your neighbours aware that there is a looming threat so they can protect themselves and call in the help for you. You will hope that the cops come soon and if possible create a more vigilant environment in future against this threat. In this case, the set of cops that hear your and your neighbour cries are resident cells of the innate immune system.
The innate immune response
The resident innate immune cells can recognize general patterns of the invading pathogen.
The first set of officers to notice that something is off are from the innate immune system 8. They include cells like neutrophils, macrophages, dendritic cells, Natural killers, etc. They are not specific to the pathogen and their activity relies on the general patterns they have learnt to recognize in the course of evolution; and inflammatory signals that come from infected cells that activate these cells.
Identifying patterns is like a cop identifying saying - "hey I know this car belongs to one of the Don Joe's guys, what is it doing in this neighbourhood. Let me call for backup and have a closer look". Or, it could be like - "oh well I know this is a gang tattoo. Send backup."
The tissue-resident immune cells are the first to recognize the general patterns of the pathogen called pathogen-associated patterns or PAMPs. For viruses, PAMPs include viral nucleic acids and patterns on some viral proteins. This is done via specialized pattern recognition receptors such as toll-like receptors 9. This activates the resident immune cells to produce cytokines (including more interferons) to call for reinforcements from the blood and lymph 10. It is worth noting that not just pathogens and cytokines from infected cells activate innate immune cells, but these cells also recognise damage associated patterns or DAMPs released when the infected cells get damaged and die 11 . Damage associated patterns are more like seeing vandalized buildings, blood, or finding a dead body.
You can sense the fight via symptoms
Following the cytokines signals, many cells of the immune system migrate to the site of infection. The newly arrived cells secrete even more cytokines to mount a full-blown inflammatory response. Some of these cytokines are endogenous pyrogens (Such as IL1)12,. The pyrogens tune the thermostat of the body (in the hypothalamus), This tuning raises body temperature causing fever. The cellular and viral machinery needed for the virus replication don't work well at higher temperatures and hence slows the virus replication. Similarly, coughing, sneezing and runny nose are also caused by molecules secreted in response to cytokines 13. Well, that's a different thing that most of the time these symptoms - coughing, sneezing and runny nose helps the virus to spread more than it helps the host in keeping the virus away from the lower respiratory tract.
101 ways to kill the pathogen
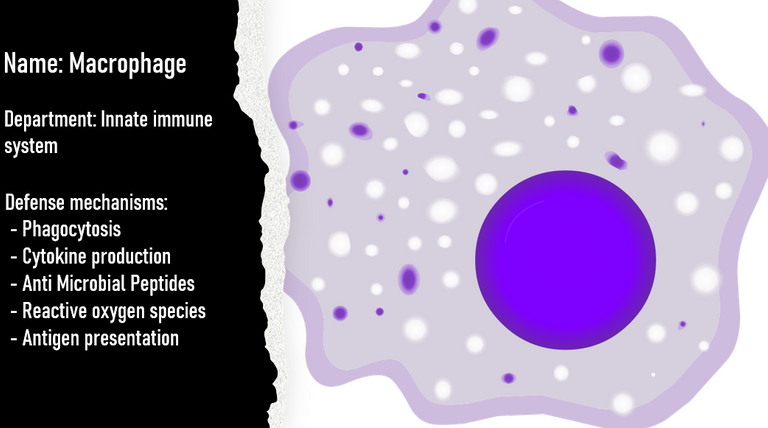
Monocytes and macrophages
Meet the macrophage
Adapted from A. Rad, Mikael Häggström, Spacebirdy, RexxS, domdomegg | CC BY-SA 4.0
Neutrophils wreak havoc, but they are like the Spiderman
Adapted from BruceBlaus | CC BY 3.0
The monocytes that come from the bloodstream differentiate into macrophages in the tissue. Apart from secreting more cytokines they also aim to destroy the virus. They do so majorly by eating up the virus and digesting its contents. They also eat up debris from the damaged and dead cells and clean up the room.
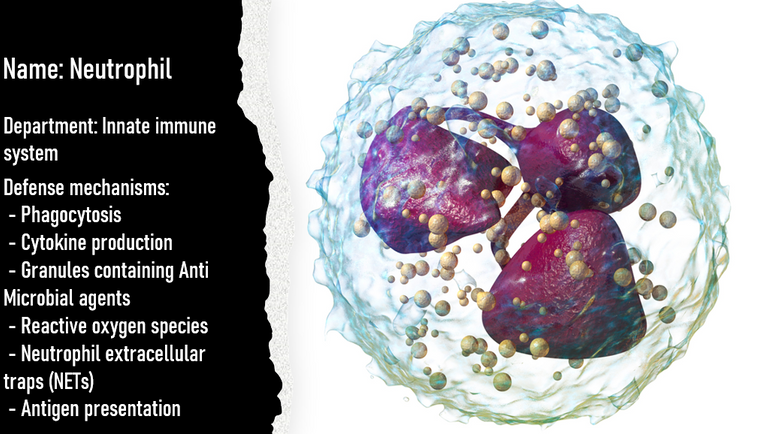
Neutrophils
Just like macrophages, neutrophils and dendritic cells that come to the site indulge in eating up the virus. But, apart from eating, these cells esp neutrophils can secrete molecules called antimicrobial peptides (such as defensins and LL37) that damage the virus in multiple ways 14. Some antimicrobial peptides don't directly damage the virus but act via changing the charge on the viral surface 15. This makes it hard for the virus to interact with the cell surface. Alternatively, they can also aggregate the virus particles and hence make it difficult for the virus to spread interact with the cells 16.
Neutrophils and macrophages also release reactive oxygen species which damages the molecules in its surroundings, including viral proteins and nucleic acid17.
Neutrophils can also release entrapment nets like a Spiderman that can trap the virus and slow down the spread.
 )
)
This terrifying looking dendritic cell is the bridge between innate and adaptive immune systems.
Drawn by @scienceblocks
Dendritic cells
Dendritic cells specialize in being the antigen-presenting cell (APC) 18. This means that they display whatever they have been eating on their surface to the T and the B cells in secondary lymph nodes, While macrophages and neutrophils also act as APCs, dendritic cells are the most potent ones and form the major bridge between the innate and the adaptive immune system. Quickly after eating up the virus the dendritic cells get drained to the lymph nodes. This is where they present the antigen to the T and the B cells. When it comes to antiviral response, a subset of dendritic cells also specialize in amplifying the interferon secretion.
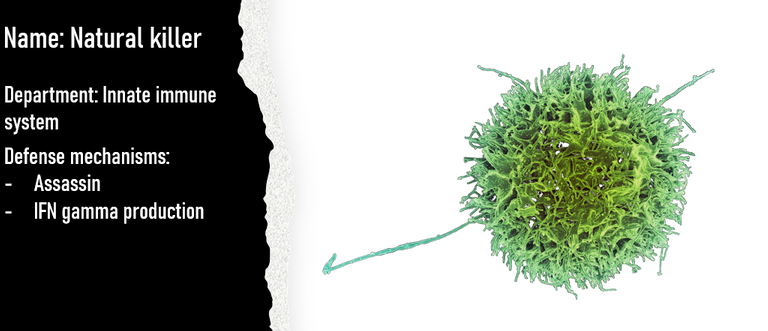
Natural Killers
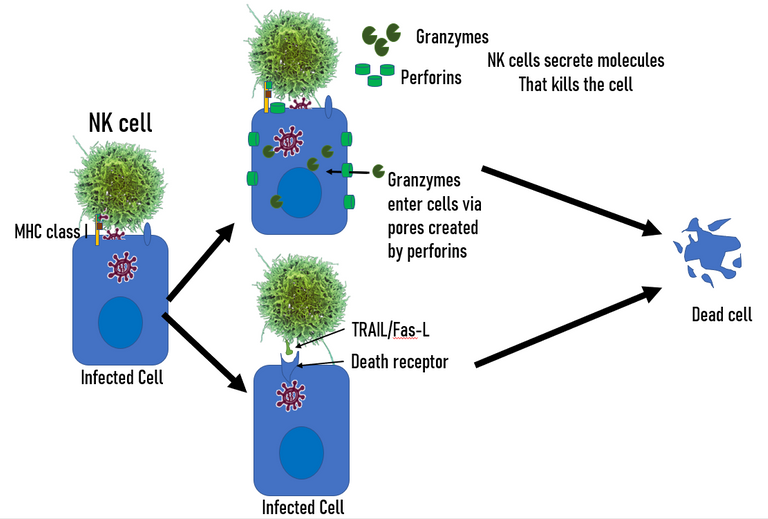
Yet another innate immune cell summoned to the site of infection is a trained assassin. It goes by the name Natural Killer cell or the NK cell. The NK cells are very sensitive to interferons present in the inflammatory environment. The interferons in the inflammatory environment along with other cytokines (IL-12, IL-15, IL-18, IL-2) recruits and activate the NK cells 19. The activated NK cell also secrete another kind of interferon called interferon-gamma. The interferon-gamma does not only potentiate the activity of NK cell but also contribute in mounting an effective adaptive immune response later. The activated NK cells once at the site starts surveillance. They can recognize the virus associated patterns on the surface of infected cells (viral molecules displayed on MHC class I of infected cells or its membrane). They can also recognize stress-related molecules on the surface of the infected cells. Once they identify an infected or stressed cell they assassinate the infected cell. They do so by secreting molecules called perforins and granzymes. Perforins makes pores in the membrane of the infected cell and granzymes enter via those pores. Once inside granzymes chew away the viral and cellular proteins in their way. NK cells can also engage with death receptors present on the surface of all cells. Once the death receptor is activated the infected cell commits suicide.
illustrated by @scienceblocks using NK cell image adapted from NIAID | CC BY 2.0
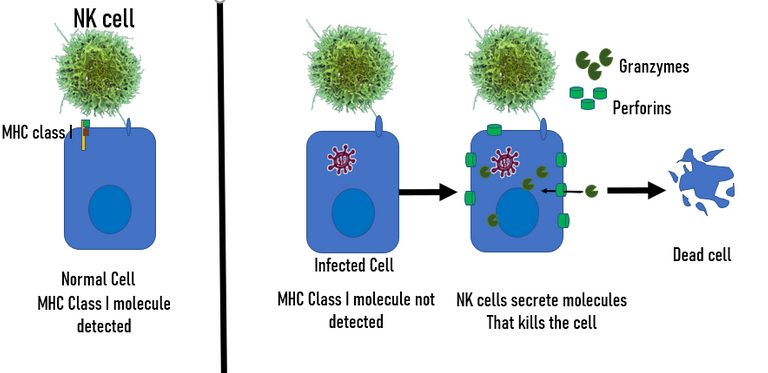
Viruses tend to reduce the expression of major histocompatibility complex (MHC class I) in the infected cells. Since, MHC is the molecule that T cell binds to check for the molecules present inside the cell, a reduction in MHC makes it hard for T cell to function. But this is not the case with the NK cells. NK cells kill the cells which don't express MHC molecules on their surface 20
illustrated by @scienceblocks using NK cell image adapted from NIAID | CC BY 2.0
Summary
Though great, none of the responses by innate immune cells are specific. Which means they can only tell that there is some pathogen in the system. They can also tell the nature of the pathogen - whether it's bacteria, fungi or a virus. They can't tell which bacteria or which virus. Even the attack they mount on the invading pathogen is not specific. The reactive oxygen species for instance doesn't know which molecule it is damaging, It damages host and virus molecules alike. Similarly, enzymes and antimicrobial peptides released by the neutrophils when in access can harm the host cells when in excess. Hence, this kind of approach is not always friendly. All the positive feedback loops can lead to excessive inflammation and cytokine storm that causes damage to the organs and even death. Hence a more trained and targeted response specific to the pathogen is desired. This is where officers of the second department called the adaptive immune system come into the picture.
Well, I did write about the adaptive immune system as well. But I realized that there is a lot of information to process until this point itself. Hence, I will be posting about it in the second episode. And then in the third episode, we will learn about the strategies and arsenal viruses have to fight back the immune system. But, can you guess what they could be? There was a hint in the article, where virus targets the ability of cells to display viral proteins on its surface. There are more places where viruses can hit to deceive the innate immune system. Hoping for some interesting answers.

References
- Molecular Pathways in Virus-Induced Cytokine Production
- The host type I interferon response to viral and bacterial infections
- Antiviral Actions of Interferons
- Interferon activation and innate immunity
- Type I Interferons Potently Enhance Humoral Immunity and Can Promote Isotype Switching by Stimulating Dendritic Cells In Vivo
- Regulation of effector and memory T-cell functions by type I interferon
- Systemic Interferon-γ Increases MHC Class I Expression and T-cell Infiltration in Cold Tumors: Results of a Phase 0 Clinical Trial
- Molecular Biology of the Cell. 4th edition.
- Pattern recognition receptor (PRRs) ligands
- Innate Immunity to Virus Infection
- Toll-like receptor signaling pathways
- Circulating cytokines as mediators of fever
- Understanding the symptoms of the common cold and influenza
- Neutrophils in respiratory viral infections
- The antimicrobial peptides and their potential clinical applications
- Interactions of α-, β-, and θ-Defensins with Influenza A Virus and Surfactant Protein D
- Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance
- Dendritic cells in viral pathogenesis: protective or defective?
- Functions of natural killer cells
- The Molecular Mechanism of Natural Killer Cells Function and Its Importance in Cancer Immunotherapy
Thanks to @swayzilla for bringing some of these questions to my notice and gave me the idea to start this series. I wanted to do such a series on immune system since quite sometime. But,if not for that question, I may have picked up another topic. Hence, as a token of thanks I will add you as 1% beneficiary to posts on this topic, since next post.

Signing off
@scienceblocks


Wow! Thank you so much for starting this. So much information to digest and I am so glad you are putting out real information out there. Thank you also for the beneficiary to this. I look forward to your series on this.
So much for the body's immune response and funny to know that the bulk of the symptoms displayed as a result of infection by covid19 is due to the body's immune system trying to shut off the virus. This is as detailed and enjoyable as a post can be. Keep them coming
What if the thing we name a virus is actually a poison?
What if that poison is electro-magnetic radiation?
How does the body react?
What would be the best defence?
Well, the word virus is indeed based on Latin word which meant poisonous secretions. That would be akin to what we now call venom. But over the course of time the word virus had taken a completely different meaning.
A virus is a piece of genetic code wrapped in some protein and lipid membrane, with one goal - to produce more copies of that genetic code. And for this it holds our cell hostage. A poison is any substance that when ingested, injected or absorbed causes illness and death. So while yeah you can loosely think of a pathogenic virus, bacteria or fungus as a poison but the usage of word poison is majorly for chemically toxic substance. Living organisms such as snakes, plants, fungus and bacteria do secrete toxic substances nevertheless. For e.g. botulism toxin, also used in BoTox come from a bacteria and is one of the most potent toxin known to mankind.
Neatly avoiding three out of four questions? Interesting.
Maybe I missed. I see I have answered your question on - virus vs poison. Radiation poisoning and response of body to it. I am not sure what you meant by last question of best defence? I will sure try to answer that too if you can elaborate.
Coming to your second question, radiation can cause poisoning. Electromagnetic radiation such as short wavelength UV, X-rays or gamma radiation cause DNA damage in organism exposed to them. The damage leads to malfunction of cells and then organs. Many people for instance died of radiation poisoning in Chernobyl post meltdown of a nuclear reactor. And yeah cells stressed with such radiation also turn on defence response. But that is a different ball game altogether and is unrelated to viruses. If someone tells you that radiation from 5g tower is toxic they would be making that shit up. Remember that for radiation poisoning you need UV and shorter wavelengths.
Nope. Electro-magnetic radiation is not quite the same thing as nuclear radiation.
You seem to have misunderstood.
The symptoms of overload of EMFs are remarkably similar to flu with one further tell-tale sign - hypoxia. Remind you of anything?
I also mentioned UV and X-rays. Well nuclear does contain other stuff - alpha and beta particles. But it also consists of gamma radiation which indeed is ionising radiation. But I realise your question was regarding the debates the EMF and EHS. My apologies for misunderstanding you. Personally, I have not come across strong and reproducible evidence for that. But who knows. I think it's best to stay away from EMF if one feels they are sensitive to it and seek an opinion of health care professional to be sure and rule out other possibilities.
For this particular case of virus how we know it is not EMF is because we have isolated the virus from patients, grown it in cell culture, imaged it under cryo EM and reproduced the symptoms in the animal models. And it's not just the original virus but also the strains that we can isolate and grow in the petridish. The visual evidence of virus and the fact that it can cause the same disease provides a strong evidence for this case. Do let me know if you think otherwise or if you think I am still not getting your question. I would be happy to hear and answer to best of my abilities.
Thanks.
So, the deceptive claim that the "virus" has been isolated rolls on unchallenged, does it?
Then may I suggest that you claim the reward as linked at the foot of my article here:
https://hive.blog/deepdive/@francesleader/viruses-are-misidentified-exosomes
Well, I have been working with viruses for over 8 years now. And I and my colleagues have isolated the virus and done the studies including cryo EM imaging. So, I find it hard to believe otherwise, since I have done the work with my hands.
Now, coming to your article. Well a virus does use the process of exocytosis. One of the ways virus gets out of the cell is by pinching the membrane of the most cell and carrying pieces of host membrane out with it. Now it's easy to confuse this with exosomes. But there is streak differences when you compare viruses vs exosomes.
Also, I tried opening the link in your post where I can claim the reward. It says 404. I would have loved to try to win that award. While your post says that we do since for money, it's not true. I have been living at income of $500 per for over 9 years now, doing nothing but science. That's how low you get paid to do academic science. Only very very tiny share of academic work actually ends up getting translated into an industrial product and end up making money for any person in academics ever. So if just proving existence of virus can pay off like 1.5 million euros much, I would try my luck 😁.
Anyhow that aside, looking forward for your inputs on what you think about my response and if you would like to talk more about it.
Many thanks for alerting me to the removal of my link from the internet however I found a copy on the wayback machine which you may wish to respond to:
https://web.archive.org/web/20210616203940/https://www.samueleckert.net/isolat-truth-fund/
I don't believe in viruses.... this makes more sense to me:
UPDATE: 08/10//2021
Finally a journalist interviews Christine Massey! Now we must make some progress xx
https://blog.nomorefakenews.com/2021/10/07/the-non-existent-virus-explosive-interview-with-christine-massey/
THE FINAL REFUTAL OF VIROLOGY - DR STEFAN LANKA
https://www.bitchute.com/video/mIYLCaRuE6hn/
https://web.archive.org/web/20210812062751/https://drtomcowan.com/only-poisoned-monkey-kidney-cells-grew-the-virus/
Thanks for the archived link. But since the link is down are they still paying though?
Contact them. I cannot advise.
Your content has been voted as a part of Encouragement program. Keep up the good work!
Use Ecency daily to boost your growth on platform!
Support Ecency
Vote for Proposal
Delegate HP and earn more
A very detailed work here ...
Very good one dear
Thanks 🙂
Alright
Just because something looks like a "viral" infection caused by a virus organism infecting cells does, not mean the viral protein has come from outside the human genome or from a separate genome.
The human genome has many dormant and polymorphic genes that can create new "viral proteins" that can be programmed by the human DNA to do all types of functions.
For instance messenger RNA can be coded specifically by the DNA and acts very similar to how you have presented the virus organism acts.
Naturally made mRNA will be carried in nanosized packages of exosomes which can express specific proteins to latch onto cells and force the production of new proteins that the messenger RNA has told the cell to make.
This looks identical to how you suggested a virus organism spreads from cell to cell growing and releasing copies of rna or dna copies of itself.
Also just because a cell absorbs genetic proteins does not mean the cell is being hijacked.
Through endocytosis and MHC domains the cell will break down these proteins or antigens and then the cell could be programmed to differentiate into an antigen presenting cell that recognizes the parts of the antigen or genetic proteins that it absorbed and broke down.
This can create all sorts of enzyme sequences and one can be for messenger RNA to send this code to other cells to make more antigen presenting cells. Another enzyme can be made to make viral proteins such as antibodies like cytokines and interferons to destroy damaged cells or help clean up and regenerate cells.
Might I add these viral proteins are mediated by exosomes also.
Interferons are added to these exosomes packets to control the cytokines and if the cell is deficient or DNA is damaged these exosomes can be mispackaged and cause uncontrolled cytokine destruction or other defective disease loops.
Most of these so-called virus organisms are simply proteins made by dormant genes from the human genome to get rid of a toxic load of antigens and free radicals; the viral proteins are not making copies of themselves to destroy the cell as a separate virus genome. This theory might seem exciting or interesting but there is no evidence to this.
If these virus organisms were real and operated the way you say then we would have no chance of survival.