T Cell Therapy - The Path to defeating Cancer
Hi buddies,
I'll open by saying that understanding how biology rules our world will play a leading role in how we fight and treat diseases that have plagued us all along. There's no arguing the fact that, our immune system performs an excellent job of keeping us alive by fighting off external invaders such as germs and viruses. However, it's your own body that is an invader in the form of cancer cells, which are experts at evading the immune system.
According to a recent study published in the journal Nature, it may be possible to train the immune system outside the body to maintain leukemia patients in remission for up to a decade. T-cells are one of the many types of cells that make up our immune system. Each T-cell specializes in recognizing and/or attacking a certain type of invader.
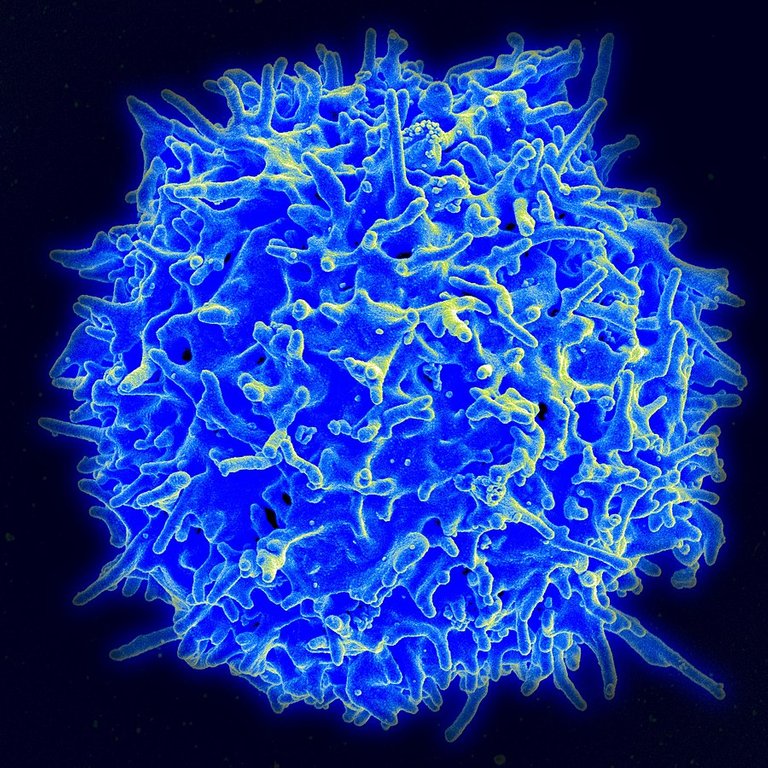
Scanning electron micrograph of a human T lymphocyte (also called a T cell) from the immune system of a healthy donor ; Source ; Public Domain
T-cells are covered in unique proteins called receptors (i.e., proteins on the surface of invaders like bacteria and viruses) that are designed to hook up with antigens in order to perform their job effectively. T-cell receptors, in other words, are more like chemical keys on the lookout for their lock. However, some illnesses, such as cancer, are masters at eluding t-cell keys. They can do this by not having enough detectable antigens on their cell surfaces, for example.
The good news is that scientists have discovered a way to extract t-cells from a patient's body, train them to detect cancer cell antigens, and then reintroduce them into the patient to fight cancer. This is known as (CAR)-T-cell therapy or CHIMERIC ANTIGEN RECEPTOR therapy. T-cell training is a sort of genetic engineering that involves a lengthy and complicated procedure. A piece of DNA is finally inserted into the T cells' genetic code, instructing them to create a specific receptor for the cancer cell antigen.

The procedure of chimeric antigen receptor T-cell therapy (CAR) is depicted in the figure above.
- T-cells (objects with the letter 't') are extracted from the patient's blood.
- The gene that codes for the specific antigen receptors is then inserted into T-cells in a lab setting.
- The CAR receptors (labeled as c) are then produced on the cell surface.
- The newly changed T-cells are extracted and cultured in the lab after that.
- After a certain time period, the engineered T-cells are infused back into the patient
Source ; License
As a result, when the freshly customized receptor latches onto its target, the t-cell calls for backup by releasing a slew of cytokines. The cytokine molecules and the army of t-cells work together to generate inflammation, which kills their targets in the best-case scenario.
While (CAR)-T-cell treatment has been approved for a few forms of cancer, little study has been done on the long-term implications. The duration of a single T-cell infusion in the body and whether the patient remains in remission are two examples of such effects. However, in 2010, two patients with chronic lymphocytic leukemia were given a single dose of CAR-T cells, and the therapy was so effective that both patients went into remission within a year. The incredible part is that they've remained that way without any extra therapy. The most recent blood work was taken a few years ago, and the team discovered that both patients still had some CAR-T cells in their blood streams.
While these are excellent findings, they do not imply that every leukemia patient will be enrolled in the program. When it first began, there were 14 patients in the cohort, however only two (2) were included in the 10-year follow-up. Furthermore, this medication is known to cause a variety of unpleasant side effects, including difficulty breathing and even seizures.
Still, further research is needed to improve CAR-T cell side effects, making it a viable choice for cancer prevention. A study published in the journal Science Advances has shown that it is feasible to assist African clawed frogs recover the upper part of their amputated limb. We humans may be able to stitch cuts together or even rebuild a significant portion of our liver, but we are not famed for our regeneration abilities. Unlike lizards and crabs, who can regrow intricate body parts such as entire limbs, we are unable to do so.

African clawed frog (Xenopus laevis) ; SourceLicense
The way it works in these animals is that their brains identify that a portion of their body has been lost, and unspecialized cells called stem cells rush to the stump and begin working. As a result, they progress from unspecialized cells to bone cells, muscle cells, nerve cells, and other types of cells. Because their bodies have already gone through the process of creating that limb, the instructions are to create a new one. We have STEM cells, too, but instead of sending STEM cells when we lose a limb, our bodies send signals to cover it with scar tissue as fast as possible so we don't bleed out or get an infection.
The researchers wanted to see if they could use 115 female African clawed frogs to trigger limb growth in humans. These animals don't have regeneration ability as adults, so if they lose a leg, it's gone for life. To test their idea, scientists chopped off a portion of one rear limb and either left the frogs to heal on their own as a control group, or covered their wounds with a small silicone cap packed with silk proteins and a 5-drug cocktail.
Each drug had a specific purpose: to reduce inflammation and the creation of scar tissue, as well as to promote normal tissue growth. The scientists had only used one medicine in a previous study, and while it did grow something back, it was more of a spike than a leg. As a result, it appeared as though there was a flash growth with the first stage of bone regrowth visible.
However, in this latest study, the caps were worn for 24 hours before the researchers observed what grew over the next year and a half. Just a few days in, the team observed that for the frogs that got the drugs, the molecular instructions their bodies had used as embryos had reactivated. And over the course of those 18 months, they grew back into the shape of a leg. While none of them resembled the frogs' other leg, two-thirds of the frogs who were administered the medications had regrown much more sophisticated tissues and leg structure. They were even able to regrow toes, but not toe bones, according to researchers. The frogs' legs could also respond to touch and be used for swimming. All of this in only one treatment day!
To conclude the blog, this research is focused on amphibians, and human trials are still a long way off. A lot more work is needed to figure out what the perfect drug cocktail needs to be to regrow the best limb possible. The prospects are promising, I envision a world where people may be able to select whether they wish to recover a missing limb or have the benefits of a robotic one decades from now. It's not impossible!
You can share your thoughts below as well! Cheers y'all
And perhaps you're interested in checking out more details, Check these links:
Decade-long leukaemia remissions with persistence of CD4+ CAR T cells
TCell
Scientists regrow frog’s lost leg
Chimeric antigen receptor T cell
Acute multidrug delivery via a wearable bioreactor facilitates long-term limb regeneration and functional recovery in adult Xenopus laevis
Posted from HypeTurf
This is amazing @mengene, I am a medical student and I am doing Leukemia topic right now, seeing such a relatable blog on hive feels so good. Keep it up, it was an informative blog.
Thanks for taking your time to read through! Glad you can relate to the blog!
My pleasure 😊
Feel free to share about your research here! We will be very happy to read from you :)
Thank you!!
I will do it for sure.
I am still perfectly waiting for the day science will provide a perfect cure to any ranges of cancer
We all are! It's been a menace, especially in this very generation of ours! Thanks for stopping by my blog!
Thanks for these very interesting stories. I must admit that I am a bit lost and do not see properly the relation between CAR T cells and the regenerative frog story. Do you mind clarifying this? Maybe this is obvious, but I don't see it. Thanks in advance.
To go back to the first topic, the findings you described look promising. It is however clear that we need to move on with further tests with more statistics before generalising this as a standard treatment. However, at least things (i.e. research) move. This is what matters most at the end of the day, in our fight again cancer.
PS: I had never seen a T-cell scan before today. I know this is a detail of your blog, but this is a cool one!
They are not related per se ... I included the regenerative frog story, since i came by it as one of the recent advances in genetic engineering as well :)
Ahah OK, we have two different stories here. Thanks! Now everything is clear :)
"Management drugs are more profitable than curable drugs when it comes to cancer. "
Just sharing one of the conspiracy theorists views about cancer and drugs. There's this belief that big pharmas have the drugs to cure cancer, but that erode the profit associated with management drugs.
It's not much of a conspiracy in my own opinion! Big Pharma has been calling the shots on who and what deserves treatment. They've got the money and power and more drugs we buy, more money they make, and more revenue they get to announce, so people keep investing in them and buying their stocks!
Congratulations @mengene! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s):
Your next target is to reach 400 replies.
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPCheck out the last post from @hivebuzz:
Support the HiveBuzz project. Vote for our proposal!
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider delegating to the @stemsocial account (85% of the curation rewards are returned).
You may also include @stemsocial as a beneficiary of the rewards of this post to get a stronger support.