THE CHEMICAL BALANCE BEHIND THE HABER PROCESS// Ammonia synthesis

Authored by @madridbg, via Power Point 2010, using public domain images.
Greetings dear members of the #hive platform, continuing with the process of socialization of chemical processes applied on an industrial scale, this time we will study the balance that must prevail in chemical reactions, for this we will focus our study on the HABER process as an industrial mechanism used in the production of ammonia.
As has been constant in my publications this type of material we will share it through the community platform @stemsocial, pioneering community on topic of scientific interest with practical contributions.
INTRODUCTION
Chemical reactions are processes that are present in all systems that make up our life, from biological, to metabolic aspects, are governed by chemical principles associated with reactions. Therefore, it is of utmost importance to know the mechanism of action of these systems, which fully answer three questions that we will describe below.
1. What happens when a chemical reaction occurs?
2. What delimits the rate of a chemical reaction?
3. How far can a chemical reaction go?
Before answering this question, it is necessary to remember and differentiate the terms chemical reactions and chemical equations, where the first is the process that takes place when the reactants interact to generate substances different from those of its initial constitution, for its part, the second is nothing more than a representation that we can make on paper and that allows us to understand the operation of the real process.
In this sense and based on the above, we can establish that the answer to the first question is not provided by stoichiometric processes, understood as the quantitative relationship between reactants and products, therefore what happens is the interaction of amounts of substance to form new products.
According to approach number two, we can establish that speed is synonymous with kinetics, which allows us to visualize the behavior of particles as a function of time. In this sense, we will leave approach number three to be answered throughout this article, which will allow us to reinforce and understand the operation of the HABER process in the synthesis of ammonia.
OVERVIEW OF CHEMICAL BALANCE
When talking about chemical equilibrium, we have to associate it with the capacity manifested by chemical processes, where the concentrations of the reactants compared to the concentrations of the products are constant over time.
An example of this process, we can visualize it when we place a container with water which we will cover and place to boil, as time passes we can observe that a set of bubbles are formed in the form of water vapor, the same try to leave the container but thanks to the lid do not achieve it and end up returning to it.
When the number of particles trying to leave equals the number returning to the vessel, we can say that we are in a dynamic equilibrium. The same happens with the reactions in equilibrium, part of the reagents are exhausted to form new products and these in turn to regenerate the reagents, there comes a point that the process does not advance since we have reached an equilibrium in the chemical reaction and depending on our needs this balance can manipulate them to the right ie formation of products or to the left regeneration of the reagents.
In such a way that when the rates of the reactions are equalized, the concentrations of reactants and products remain constant and the system has entered equilibrium. At the biological level this behavior is fundamental, as an example we can cite the transport of oxygen by hemoglobin, where the reaction must be in equilibrium or our system would collapse due to lack or abundance of oxygen to the different organs that make up our body.
The process responds to the following equation:
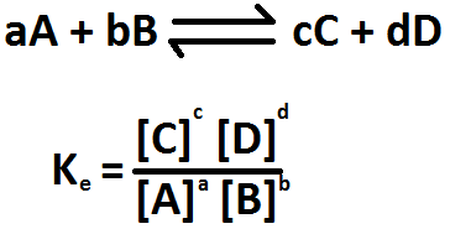
Fig. 2. Representation of the equilibrium constant. Author: Norabermudez
If we analyze the equation, we realize that to achieve equilibrium, we must relate the concentrations of the products as a function of the concentrations of the reactants, which will allow us to determine the equilibrium constant of the system. In the same way we must take into account that in its majority all chemical reactions tend to make reversible.
BEYOND THE EQUILIBRIUM CONSTANT
In the previous section, we did a previous review on the equilibrium constant, where we could observe that this depends on the equilibrium concentrations of both reactants and products, where the same variable that today we call equilibrium counters is only found to be constant when the temperature of the system does not change.
In this sense, through the equation we can predict different behaviors of a chemical process, thanks to the dynamism and interpretation that can be given to the quantitative representation. In such a way, through it we can observe the direction of the chemical equation.
On the other hand, the same equation allows us to determine the concentrations of the substances in equilibrium from the equilibrium constant an example of this process can be determined in the following statement:
Proposed exercise:
The formation of trans-stilbene is carried out by cis-stilbene at a concentration of 0.850mol/l. if the process is carried out in at 200°C and the equilibrium constant is 24. Determine the concentrations of cis- and trans-stilbene.
To answer these questions we must follow the following sequence.
Step 1. Extract the data provided by the statement.
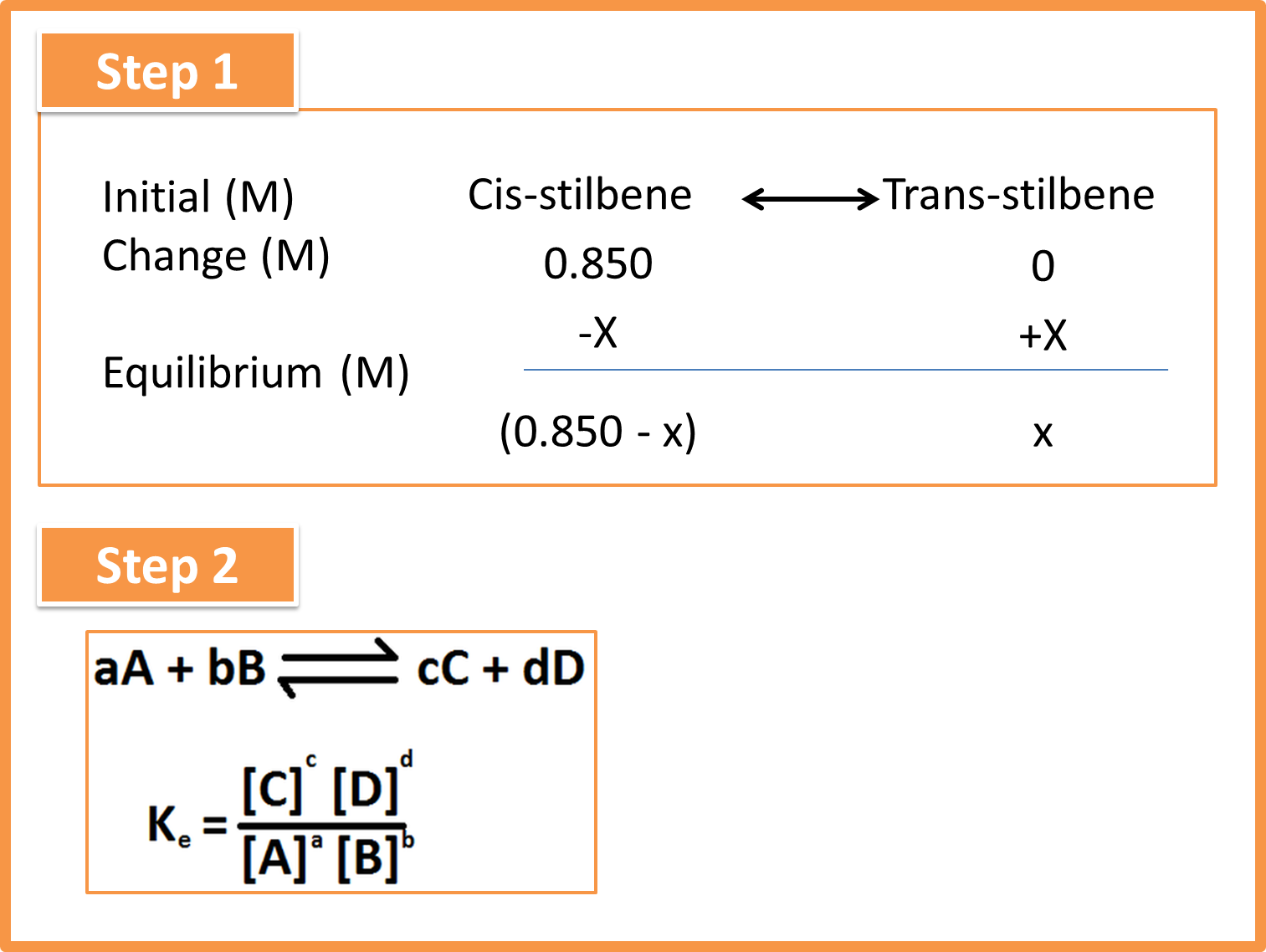
Fig. 3: Steps 1 and 2 in solving the problem. Author: @madridbg, via Power Point 2010.
Step 2. Locate the path or way that allows us to reach the solution of the problem, in this case it will be the equation that corresponds to the sequence in equilibrium.
Step 3. Substitute the values and apply the corresponding mathematical processes.

Fig. 4: Steps 3 and 4 in solving the problem. Author: @madridbg, via Power Point 2010.
As we can see in the images above, by means of the equilibrium equation we can determine the concentration of the participating substances.
THE HABER PROCESS AS A TECHNOLOGICAL MECHANISM ASSOCIATED WITH CHEMICAL EQUILIBRIUM
At the industrial level it is of utmost importance to know the factors that affect the equilibrium of a reaction, since the manipulation of these allows us to obtain effective results on the process being developed. In this sense, the Haber process is the one used in the synthesis of ammonia, which allows a chemical reaction between hydrogen molecules and gaseous nitrogen, a reaction that is catalyzed by heterogeneous accelerators.
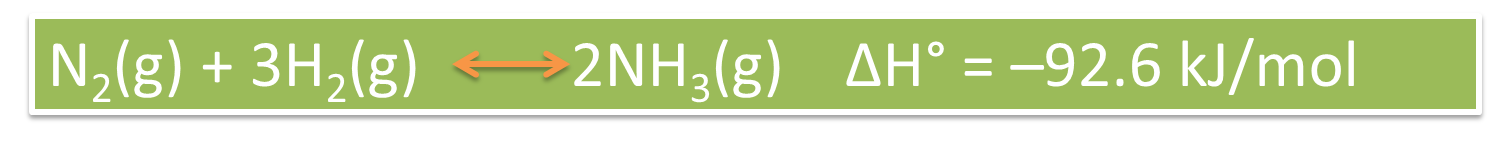
Fig. 5: Chemical equation representing the synthesis of ammonia. Author: @madridbg, via Power Point 2010.
If we analyze the equation we can establish by stoichiometry that 1 mole of nitrogen reacts with 3 moles of hydrogen and produces 2 moles of ammonia, however, if we observe the variation of the enthalpy, we realize that the reaction is exothermic so it takes place with surprising speed which decreases the efficiency of the process.
Now if we want to be efficient in the production of ammonia, we should manipulate the temperature variable and use the equation at low degrees Celsius, which is proven to generate a more efficient process in performance, but slower in obtaining, which ultimately shifts the balance of the reaction to the sides of the reactants. Thus, at industrial level it is preferred to work with the fast mode, i.e., raising the temperature, since the quantities obtained per hours of production are greater and the displacement of the equilibrium tends to be on the side of the products.
So we can conclude that it is preferable a fast commercial production knowing that it will generate us a higher cost in the process, for this reason the technological process is carried out at high temperatures and high pressures which allows us to generate ammonia on a large scale.

Fig. 6: Graph demonstrating the temperature-yield relationship in the production of ammonia. Author: @Chang, (2010).
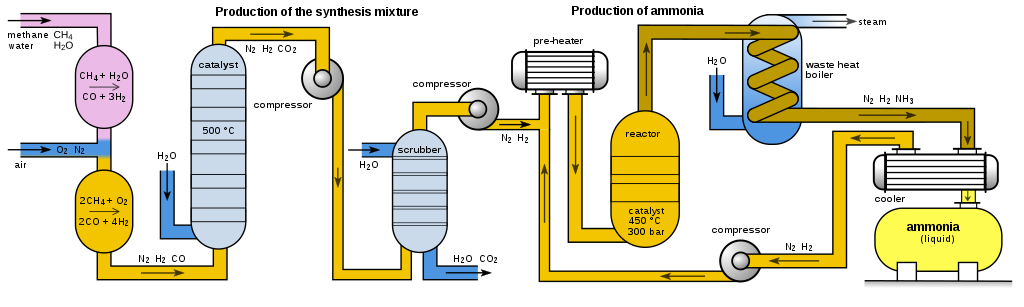
Fig. 7. Representation of the HABER process. Autor: User:Sven
CONTRIBUTIONS FROM THE TOPIC
Throughout the development of this publication we were able to address what is related to the equilibrium constant that affects chemical reactions, likewise we were able to understand the influence of chemical kinetics on the behavior of the equilibrium, as well as we could apply quantitative processes to determine the concentrations of the substances involved in a chemical reaction.
CONSULTED BIBLIOGRAPHY
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] Estupiñan Silva, Rosmerlin Quesada, Benjamiín. (2010). The Haber-Bosch process in the agroindustrial society: danger and alternatives. Latin American Council of Social Sciences (CLACSO).. Artículo: Acceso Online
[3] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[4] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[5] WADE,LEROY. (2011). . QUÍMICA ORGÁNICA. VOLUMEN 2. SÉPTIMA EDICIÓN. PEARSON EDUCACIÓN, MÉXICO, 2011 ISBN: 978-607-32.()793•5. ÁREA: CIENCIAS
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
#posh twitter
https://twitter.com/BGMadrid/status/1387523928004890629?s=20
Your post has been voted as a part of Encouragement program. Keep up the good work!
Use Ecency daily to boost your growth on platform!
Support Ecency
Vote for Proposal
Delegate HP and earn more