ENZYMATIC ACTIVITY// COACTION OF VITAMINS AND COENZYMES IN CATALYZED METABOLIC REACTIONS

Author: @madridbg, via Power Point 2010, using public domain images. S. Hermann & F. Richter
Greetings dear members of the #hive platform. Continuing with the process of socialization of topics of practical interest associated with chemistry in its different branches, in this installment we will be addressing the enzymatic functioning, focusing our attention on the functions performed by vitamins throughout this process.
As has been constant in my thematic approach, this publication will be shared through the @stemsocial community, who have maintained a position associated with the support of scientific content.

INTRODUCTION

The metabolic system in its design is a complex but specialized process where protein systems play a very important role, such is the case of enzymes, the same as biological catalysts exhibit extraordinary efficiency when compared to inorganic processes that many of us know.
That is why all cells have been arranged with hundreds of enzymes ready to receive an order and fulfill the function assigned to them that allow the development of life. At the metabolic level, enzymes are specialized substances that accelerate chemical reactions, so it is assumed that these substances decrease the activation energies of the different processes carried out in the human body.
Although there are a great variety of these substances, they have been programmed in such a way that they can only develop an activity that is assigned to them, hence the specificity of these substances and the behavior they assume.
In this sense, through this publication we will focus our attention on describing the enzymatic behavior depending on the natural cofactor or companion of the same, which will allow us to have a broader vision about the enzymatic functioning.

ENZYME GENERALITIES

In the previous section, we mentioned that enzymes are biological catalysts of protein nature, which fulfill a vital function and the same is associated with the increase of the speeds of the processes, equivalent to 1x103 to 1x1020 which in normal conditions in many cases would not start.
If we analyze the term enzymes, we realize that its name comes from a Greek word meaning yeast, it was coined at the end of the 19th century when the fermentative compartment of sugars was studied by the action of yeast.
In this sense, for practical reasons and convenience for this writing, we will focus our attention on the generalities associated with enzymatic specificity, where the substances act according to the substrate involved, so that we can establish that they are substances that present a selectivity to different compounds and their behavior depends on it.
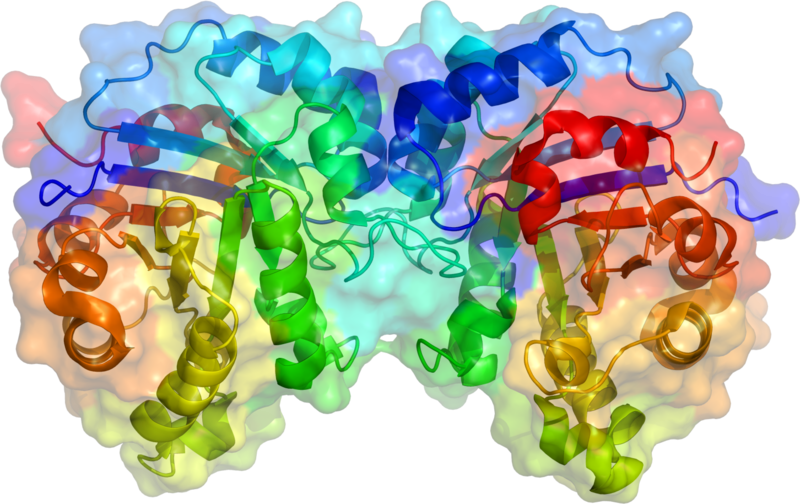
Fig. 2. Structural representation of the enzyme triosephosphate isomerase. Author: AzaToth
The aspect associated with specificity is fundamental since in a certain way it avoids large-scale complications at the metabolic level, in the first instance because it produces a considerable saving of energy at the cellular level, which allows an optimal functioning of the cell, on the other hand, prevents the generation of toxic products as intermediary of the metabolic reactions, so we can establish that they are highly coordinated process by our metabolism, hence the efficiency and complexity of the same.
We have to keep in mind that enzymes act analogously as a key (substrate) with its lock (enzyme), hence the principle of specificity, because if the lock is not supported by the appropriate key, it simply does not work, this same principle is applied at the enzymatic level, where the activity only occurs if it has the ideal conditions to function, ie, in the presence of the specific substrate.
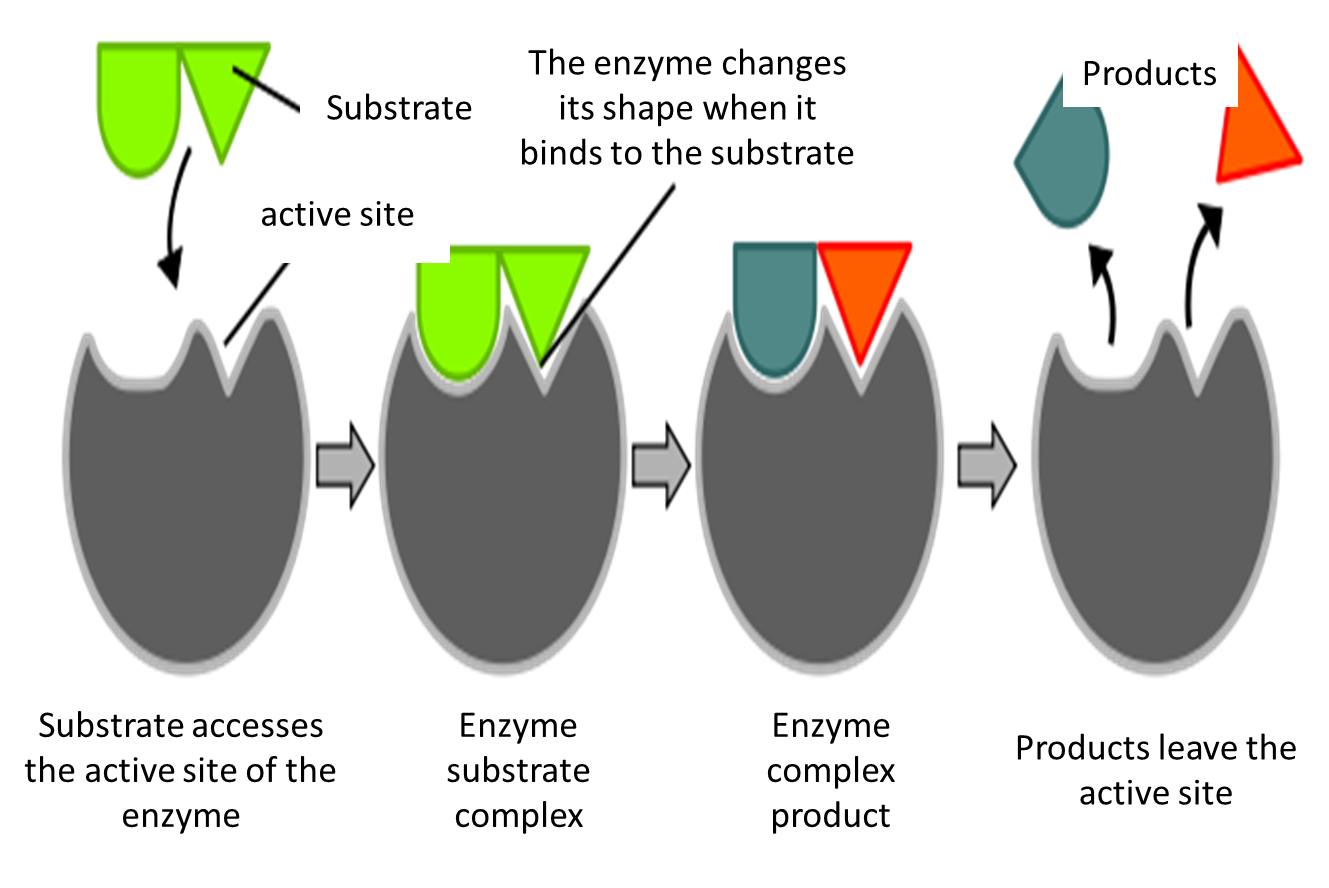
Fig. 3. Enzyme lock-key model, enzyme specificity. Author: Induced_fit_diagram.svg:
Another characteristic of enzymes is that beyond accelerating reactions, they allow combining and separating substances, in addition to the coupling of reactions that in many cases tend to become irreversible, so that if these processes are maintained over time, life would not be possible.
Therefore, enzymes allow the energy gained in one reaction to be used as a starting principle in the next reaction, a typical example of this process occurs in the hydrolysis of ATP, which is often coupled to less favorable metabolic reactions and allows both reactions to flow with great ease.

ENZYME CLASSIFICATION ACCORDING TO THEIR SPECIFICITY

In this section of the subject, we will make an enzymatic classification according to the substrate and activity developed by the enzyme, therefore these specialized substances are described according to the principles established by the International Union of Biochemistry and Molecular Biology (IUBMB).
So we will begin by addressing what concerns the oxidoreductase enzymes, which are characterized by promoting the development of those chemical reactions associated with redox processes, i.e. oxidation and reduction reactions. A metabolic example of this type of process is the one that takes place in glycolysis when lactate is transformed into pyruvate, by the action of the dehydrogenase enzyme.
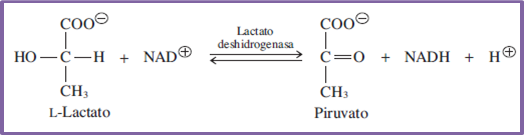
Equation 1. Behavior of lactate dehydrogenase enzyme. Author: @madridbg, via Power Point 2010.
On the other hand, the processes associated with the transfer of one or several groups of atoms or molecules are carried out through the collaboration of the transferase enzymes, which require a fundamental coupling that is usually generated by the coenzymes, which form covalent bonds that allow a group transfer according to the metabolic needs or requirements.
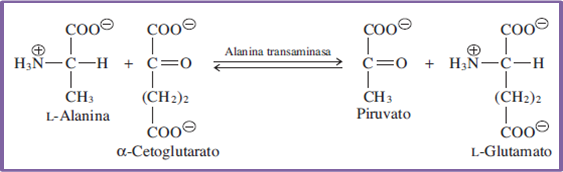
Equation 2. Representation of processes catalyzed by enzymes of the transferazyme type. Author: @madridbg, via Power Point 2010.
Otro tipo de enzima, asociado con la trasferencias de sustancias lo representan las enzimas hidrolasas, con la diferencia de que la actuación de estas, va en función de la presencia de agua, ya que esta última se comporta como un aceptor de los grupos transferidos. De igual forma podemos encontrar las enzimas liasas, estas se caracterizan por la adición de grupos o sustratos a un dobles enlace, de modo que la reacción que se lleve a cabo termine siendo de eliminación, no hidrolíticas y no oxidantes.
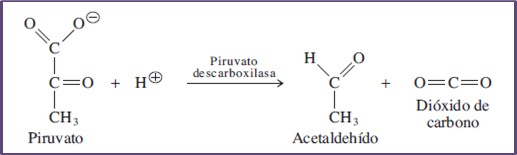
Representation of the enzymatic action of pyruvate decarboxylase. Author: @madridbg, via Power Point 2010.
Similarly we find the isomerase enzymes, which are associated with structural changes in the same molecule, are simple reactions since they are generated by the presence of only one substrate and consequently only one product is obtained, without concerns about the formation of intermediary substances.
Last, but not least, we have the l ligase enzymes, as their name indicates, catalyze the binding and union of substrates, a process that needs potential energy to be executed, and it is there, where ATP is invested, which will be restored throughout the process.
Undoubtedly, the aforementioned classification represents a generality of the process and within each category there is an endless number of enzymatic compounds that fulfill essential functions in our metabolism.

ROLE OF COENZYMES IN ENZYME FUNCTION

In order to have a clearer understanding of enzymatic functioning, it is necessary to take into account the role played by vitamins and coenzymes. In this sense, and depending on the research, in this section we will focus on the exclusive study of coenzymes, which represent a repertoire of specialized substances that fulfill metabolic functions.
Thus, in order to function, enzymes need the presence of cofactors, substances that give biological activity to inactive proteins. These cofactors can be of two types metal ions and coenzymes respectively.
The former are essential ions associated with the metallic elements that we know, among which magnesium, iron, among others, stand out, while the coenzymes are organic groups that end up forming part of the active groups where they must coexist with the substrate, here we can find the co-substrates and prosthetic groups.
These substances, function as a transfer system, as they are responsible for accepting or donating chemical groups, some donate hydrogens, others carry covalently bound chemicals.
In this sense, we can establish that co-substrates are regenerated over and over again throughout a catalysis, so we can establish that they are recyclable substances, unlike these prosthetic groups remain attached to the enzyme during the reaction. In some cases, the prosthetic group is covalently bound, while in other cases it is firmly attached to the active site by many weak interactions.
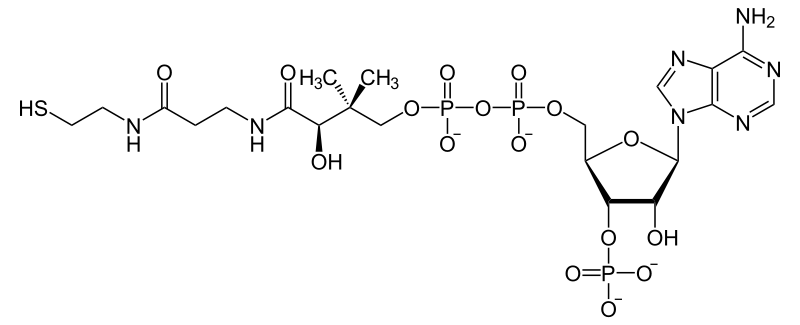
Fig. 4. Representation of coenzyme A. Author: NEUROtiker

VITAMINS C AND THEIR BEHAVIOR AS COENZYMES

Throughout our lives we have been bombarded with the information that it is necessary and essential to consume vitamins, but at some point have you ever questioned why?
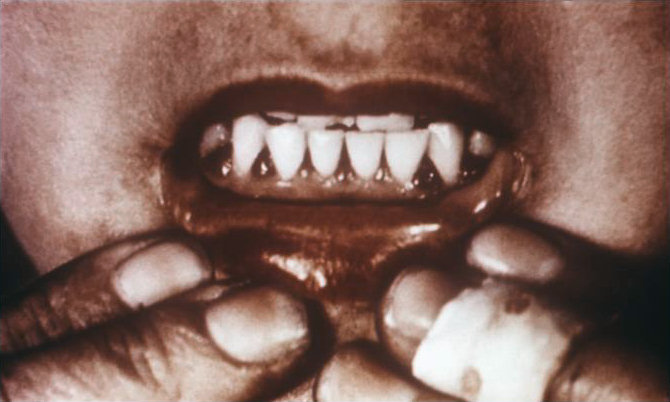
To answer the above question, it is appropriate to go back to the 1930s, when ascorbic acid was isolated for the first time, becoming the simplest and most essential vitamin to attack scurvy, a disease that manifests itself with bleeding of enzymes, skin blemishes, among others.
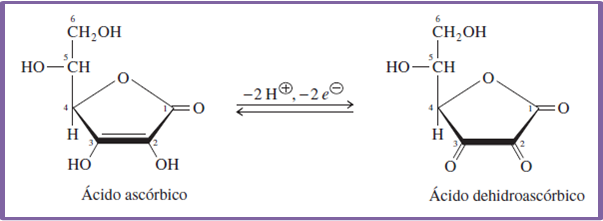
Fig. 5. Scurvy, a disease associated with vitamin C deficiency. Author: Wikimedia Commons
These substances become essential because our organism is unable to produce it, hence it must be consumed through an external diet. Chemically, ascorbic acid is a lactone type carbohydrate, that is, an intermediate ester where the carboxylate group of carbon 1 is condensed with the hydroxyl group of carbon 4, forming a ring structure.
Based on the above, it can be inferred that vitamin C does not meet the above-mentioned characteristics of coenzymes, on the contrary, its behavior is associated with the reducing hydroxylation groups of collagen.
Equation 4. Representation of ascorbic acid in the middle of a redox reaction. Author: @madridbg, via Power Point 2010.
Thus, it is necessary to clarify that not all enzymes accompany enzymatic processes, but those processes that depend on the presence of these enzymes cannot develop in their absence, hence the importance of an adequate intake of vitamins, via food supplement or through our diet, which will allow the proper functioning of our metabolism.

CONTRIBUTIONS OF THE SUBJECT

Through the present publications we were able to approach a set of characteristics behind the enzymatic processes, which allowed us to know the behavior of vitamins and coenzymes in the development of our metabolism, at the same time, it allowed us to approach an enzymatic classification, depending on the type of reactions that these catalyze, leaving greater clarity throughout the process.

BIBLIOGRAPHY CONSULTED

[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] McMURRY E., John y Fay C., Robert. (2008). General Chemistry. Fifth edition PEARSON EDUCACIÓN, Mexico, 2009 ISBN: 978-970-26 1286-5.
[3] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). GENERAL CHEMISTRY. Eighth edition. PEARSON EDUCACIÓN. S.A., Madrid.
[4] Sánchez-Lara Eduardo. (2014). Hemoglobin: an example of bioinorganic chemistry. Master's Degree in Chemistry. Chemistry Center. ICUAP. Meritorious Autonomous University of Puebla.Artículo: Acceso Online
[5] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCACIÓN, MEXICO, 2011 ISBN: 978-607-32.()793•5. ÁREA: CIENCIAS

OF INTEREST

For more information related to the areas of science, technology, engineering and mathematics, feel free to visit #stemsocial and #stem-espanol, communities that promote scientific advances in these areas.

0
0
0.000
#posh twitter:
https://twitter.com/BGMadrid/status/1467663336594710534?s=20
https://twitter.com/BGMadrid/status/1467663336594710534
The rewards earned on this comment will go directly to the person sharing the post on Twitter as long as they are registered with @poshtoken. Sign up at https://hiveposh.com.
Η ανάγνωση για enzyme action, cellular energy saving και lock-and-key specificity είναι εξαντλητική! Χρειαζόμουν μια αλλαγή με μηδενική βιοχημική ανάλυση. Ένας φίλος μού σύστησε το https://winhero.gr και με ενθουσίασαν οι εξαιρετικές προσφορές για την Ελλάδα. Δοκίμασα το 'Crazy Time'—είχα συνεχείς απώλειες, αλλά ρίσκαρα λίγο περισσότερο και πέτυχα ένα απίστευτο κέρδος. Αυτή η νίκη ήταν ο καλύτερος συντονισμός της ημέρας.