BACTERIAL ENERGY// Geobacter and electrochemical energy production
(Edited)
Author: @madridbg, through Power Point 2010, using public domain images.
Welcome my dear readers of the #Hive community, in this opportunity we will continue sharing scientific content associated with the usefulness of chemical principles. Content that we will share through the @stemsocial community. In this sense, we will chemically substantiate what refers to the generation of energy through Geobacter bacteria and its possible implementation in large-scale processes.
INTRODUCTION
The obtaining of electrical energy has been a subject of utility and practical importance in the life of man, which has been evolving and has become dependent on it, so that in a way we have been forced with the passing of time, to seek new techniques and more efficient processes that allow the obtaining and use of electrical energy in our daily lives.
However, despite technological advances, energy production has somehow caused an alarming deterioration in our planetary systems, since in the generation process large amounts of pollutants must be emitted, which is why new alternatives are sought daily to mitigate such alarming situation.
In this sense, research is currently being carried out to use the electrochemical energy produced by bacteria, through their internal respiration, which when in contact with elements such as iron and manganese produces a constant flow of electrons that can generate small electric currents. Process that we will describe in the body of this publication approaching it from a chemical approach.
ELECTRIC ENERGY AS A FUNCTION OF CHEMICAL ENERGY
Chemically, electrical energy, is considered as the flow or exchange of charges, where negative charges or electrons travel through an electrical conductor (wiring), as a consequence of the difference in potential that generators produce at the end attached to it. At present, the obtaining of this type of energy is variable, since we can obtain it through wind, hydraulic processes or through the burning of fossil fuels.
However, some of these processes tend to generate a negative environmental impact, which is why the processes associated with electrochemistry are an alternative to the old generation systems.
Under this conception, electrochemical energy, is understood as a process through reduction and oxidation reactions, also known as redox reactions, where the energy released in the reactions are transformed into electrical energy more efficient processes and with less impact to our planet.

Fig. 2. Exemplification of electrochemical processes. Author: Clker-Free-Vector-Images
ELECTROCHEMICAL ENERGY CELLS
In this section of the publication, we will make a descriptive tour on some of the cells with practical utility in the generation of electrical energy, from processes involving chemical fundamentals. Among those that stand out: galvanic cells and combustion cells, these will allow us to explain in detail the electrical behavior of bacteria of the Geobacter. genus.
In this sense and in references to the galvanic cells, it is based on the generation of electrical energy through oxidation-reduction reactions, its name is attributed to scientists Alessandro Volta and Luigi Galvani, who were the first to create and use this type of instrument. Chemically, the process is based on causing a transfer of electrons in oxidation and reduction systems separated from each other, but linked by a wiring that conducts electrons from one system to another.
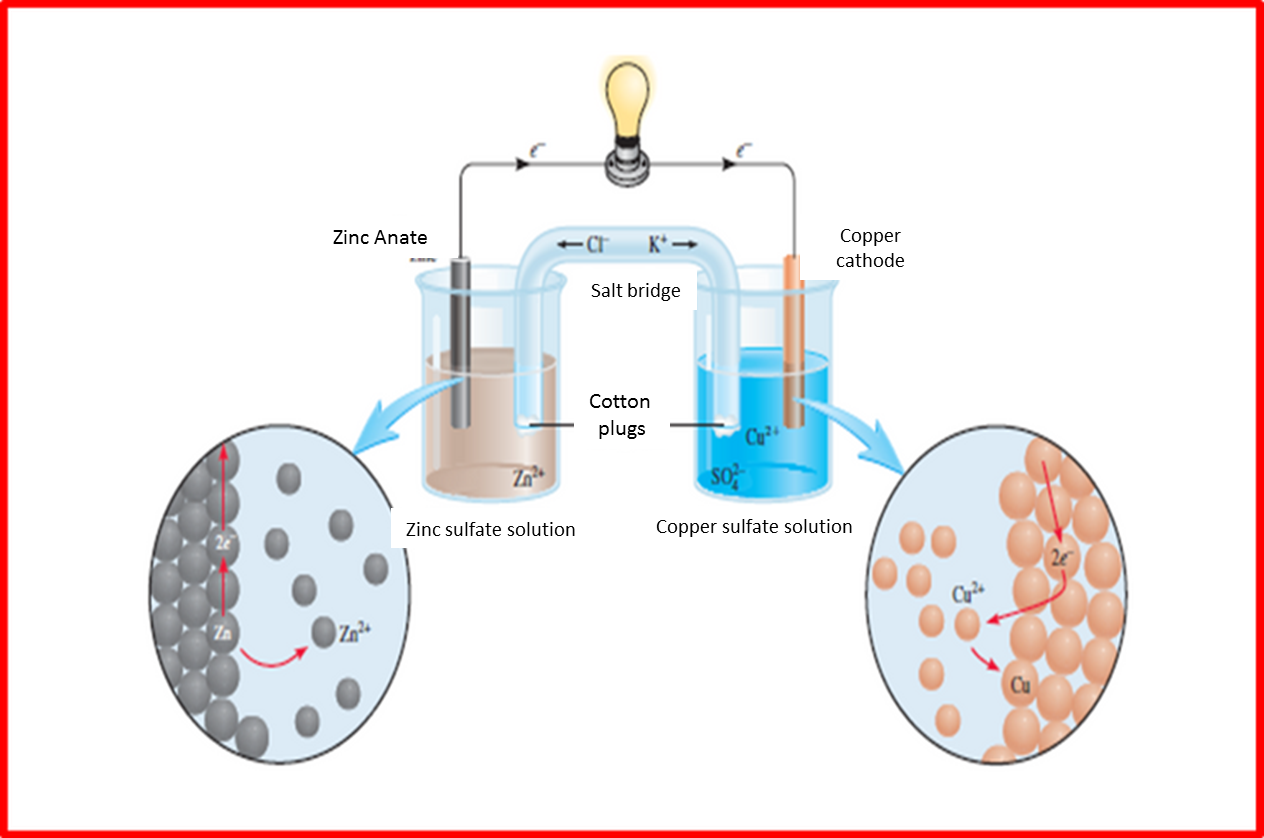
Fig. 3. Operation of a galvanic cell. Author: @madridbg, using Power Point 2010. Adapted from Chang (2010).
If we analyze the system we can observe that on the left side occurs the corresponding oxidation reaction, where the metallic zinc is oxidized to zinc sulfate, on the other hand the right side represents the reduction reaction, where the metallic copper is reduced to copper sulfate, generating the flow of electrons through the wiring that is observed on the surface of the system and that is responsible for the ignition of the bulb.
For its part, the fuel cells, is essentially a galvanic cell, so it needs a constant flow of reagents to keep running the reaction generated, is performed so that obtaining electrical energy is more efficient because in the combustion reactions that are carried out by burning fossil fuels, only 40% of the energy produced in the rest is lost as heat.
The inefficiency of this type of process is attributable to how complex it can become, because the substance is burned to generate water, which must be transformed into steam by the heat generated. Steam that allows the operation of the turbines that will make the conversion of heat energy into electrical energy.
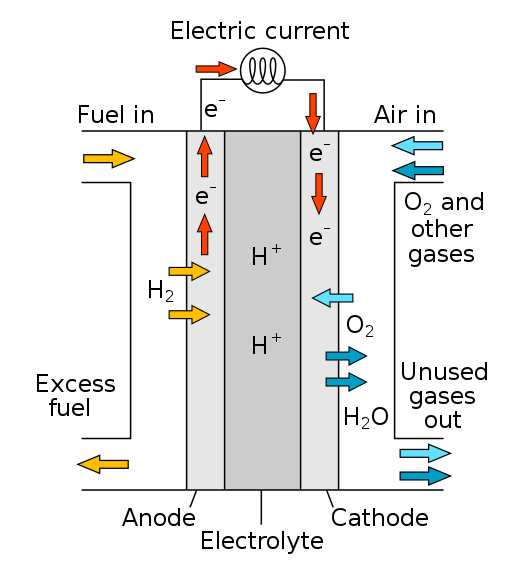
Fig. 4. Fuel cell operation. Author: R.Dervisoglu
MECHANISM OF ACTION OF THE BACTERIA Geobacter
The emergence of the Geobacter bacterium is attributed to researchers at the University of Massachusetts, led by Dr Derek Lovley. Who through studies conducted to the Potomac water in Washington found this species of bacteria.
The same are attributed a variety of uses, among which may be mentioned, the ability to produce natural magnets in environments with deep sedimentation also has been used in the degradation of organic substances present in oil by anaerobic action, also has the ability to convert harmful substances such as uranium in a neutral component.
Undoubtedly, these are extraordinary applications for this type of organism, however, to respond to the guidelines of this publication, we will focus on the ability to produce electrical energy from electrochemical reactions carried out by this tiny organism.

Fig. 5. Representation of the Bacterium Geobacter. Author: qimono
Structurally, its body is adapted with long tentacles that behave like a cable or conductor through which electrons travel and where it causes the oxidation of the substances it attacks, usually tends to do iron oxide.
Scientists led by Lovley took advantage of the species' extraordinary ability and developed a kind of bacterial combustion cell using graphite electrodes. On this surface the bacteria grow naturally, creating a kind of biological membrane that responds to the following equation.

Fig. 6. Representation of organic matter decomposition. Author: @madridbg, via Power Point 2010. Adapted from Chang. (2010)
If we analyze the equation, the acetate ion represents the organic matter that through the degradation of this, electrons are released which are available to the bacteria located at the anode of the system, then transferred to the cathode through the graphite rod and from there to the electron receiver which for this particular will be oxygen. This allows the generation of small electric currents, although the energy generated so far is small, it can become an alternative to abandon the consumption of fossil fuels that threaten the biotic systems of our planet.
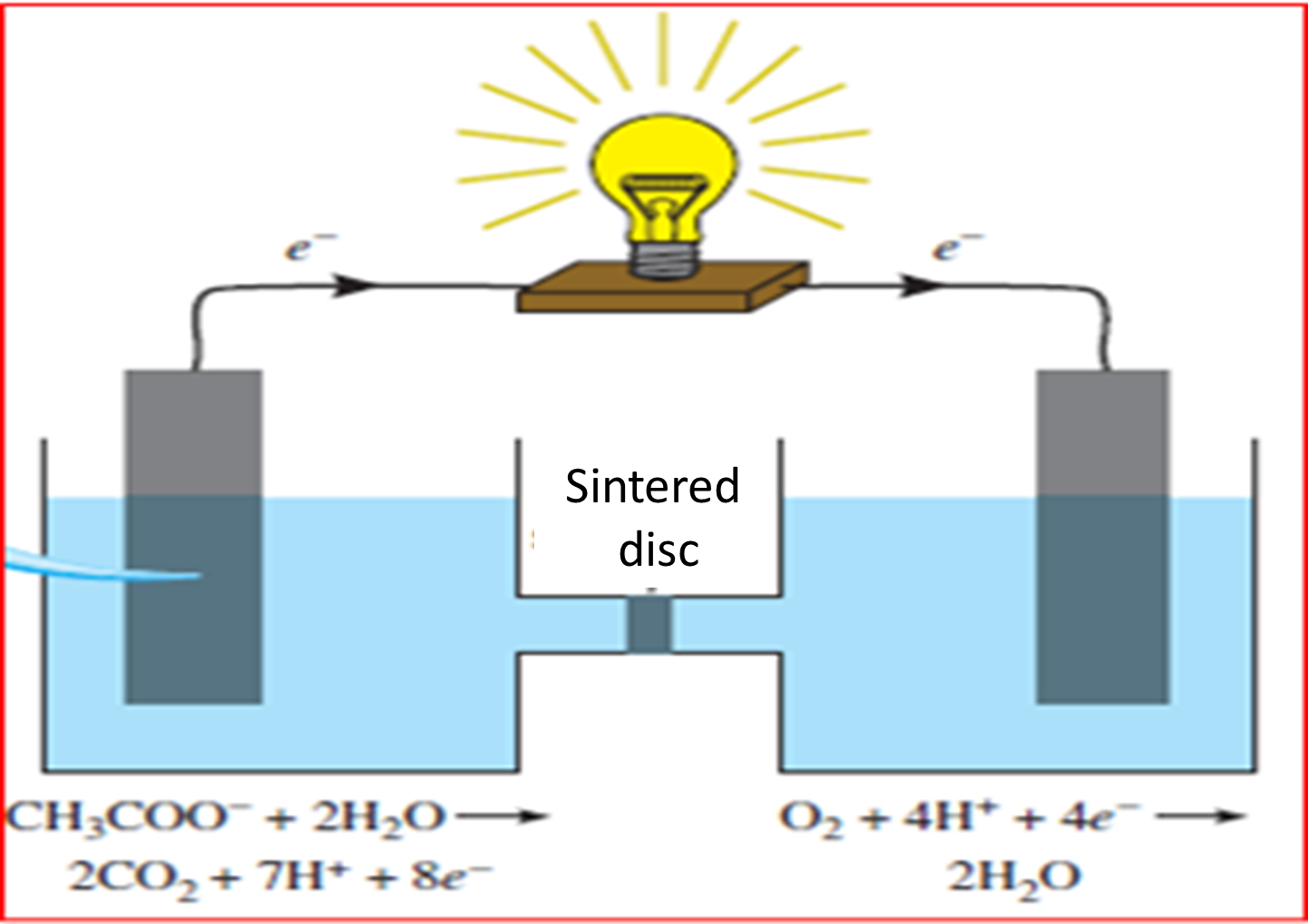
Fig 7. Energetic functioning of Geobacter bacteria. Author: Chang. (2010).
THEMATIC CONTRIBUTIONS
Through this publication we were able to address some of the benefits that nature offers us, in this case the Geabacter bacteria is presented as a mechanism that in the long term can be an alternative in the generation of electricity without having harmful effects on our environment. In the same way, the behavior of the species could be chemically approached, where they were justified from the scope of chemistry.
BIBLIOGRAPHY CONSULTED
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[3] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[4] Romero Alba, Vásquez Jorge y Lugo Armando. (2012). Bacterias, fuente de energía para el futuro. Tecnurra V. Vol. L. Article: Online Access
[5] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCATION, MEXICO, 2011 ISBN: 978-607-32.()793-5.
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/BGMadrid/status/1367186084837351434
#posh twitter:
https://twitter.com/BGMadrid/status/1367186084837351434?s=20
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.
HTTP is in use instead of HTTPS and no protocol redirection is in place. Be careful and do not enter sensitive information in that website as your data won't be encrypted.
It's also a good habit to always hover links before clicking them in order to see the actual link in the bottom-left corner of your browser.
This auto-reply is throttled 1/20 to reduce spam but if it still bothers you reply "OFF HTTP". Or reply REVIEW for manual review and whitelisting.