APPLIED CHEMISTRY: Sodium azide a useful compound to save life, chemical rationale
(Edited)

Author: @madridbg, through Power Point 2010, using public domain images.
Welcome my dear members of the #Hive platform and especially those who make life daily in the @stemsocial community. Let the present publication serve to continue with the process of socialization of applied chemistry, where we will address what concerns the sodium azide compound, implemented in the air bags of automobiles and that have allowed to save hundreds of lives since its implementation.
INTRODUCTION
The technology applied to automobiles has been evolving and every day it becomes more sophisticated, nowadays these incredible machines are equipped with motion sensors, webcam, gps, video and audio system, among others. Tools that have been adapted with the comfort of the occupants in mind.
The safety implemented to protect the lives of people has not lagged behind, today we see cars that have airbags device, also known as airbags, which are activated at the precise moment of an impact above 30 km / h and have saved countless lives.
This system is a complement to seat belts and provides greater protection to vehicle occupants. Therefore, through the publication we will address the chemical operation implemented in this mechanism and quantitatively demonstrate the amount of sodium azide necessary for the airbags to activate at the right time and that allows it to fulfill the function of saving life. Similarly, we will take a basic tour of the chemical principles applied in the system.
THE STOCHIOMETRY OF REACTIONS FROM THE VOLATILITY OF GASES
When talking about stoichiometric reactions, we usually associate them with solid or liquid substances, however, gases also respond to these quantitative calculations and allow us to determine the amount needed for a chemical process to take place.
In that sense, in this section we will describe the stoichiometric principles implemented in the ideal gas equation, where we can address variables such as: amount of substances in moles or grams, temperature and pressure of the system.
For this we must take into account, that the ideal gas equation results from the combination of three fundamental laws that were implemented by Robert Boyle, Jacques Charles and Amedeo Avogadro respectively. The first established the relationship between volume and pressure of the substance and established that these variables are inversely proportional, i.e., if the system shows an increase in volume, the pressure of the system would decrease.

Fig. 2. Representation in motion of Boyle's law. Author: NASA's Glenn Research Center
Referring to the second, he established the relationship between the variables volume and temperature, where it is stated that these variables are directly proportional, in other words, as the volume increases, the temperature increases.

Fig. 3 Representation in motion of Charles' law. Author: NASA's Glenn Research Center
In reference to the third one, he made a correlation between the variables volume and amount of substance and established that the volume of different substances are directly proportional to the mass of the individual molecules.
Under the above conceptions arises the equation of ideal gases where it is handled that:
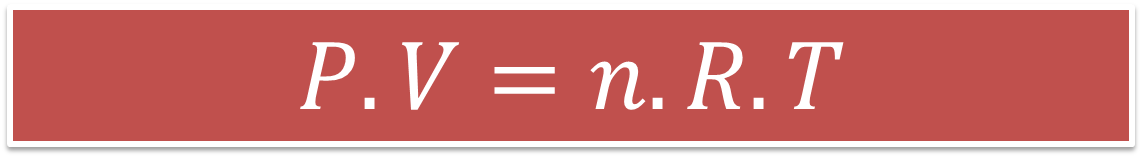
Equation 1. Equation that demonstrates the ideal gas law. Author: @madridbg, through Power Point 2010.
Where (P), represents the pressure, (V) the volume, (n) the amount of substances in grams or moles, (R) the gas constants and (T) the temperature of the system.
KINETIC THEORY AND THE DISPLACEMENT OF GASES
If we analyze the velocity of gases from the kinetic theory, we realize that the particles of these substances, move at an approximate speed of 1500m/s which allows inferring that gases have the ability to move over long distances, however, this is not fulfilled, since product of the collision between molecules this speed is reduced, as well as the change of direction of the same.
In this sense, it is necessary to address the principles of diffusion and effusion of gases, which allow us to understand the mechanism of action of air pockets. In reference to diffusion, it is understood as that migratory process that exerts the molecule as a result of the set of random collisions that occurs in the container containing the gas, if the process is established between two or more gases a homogeneous mixture is generated in the system as the particles intermingle with each other.
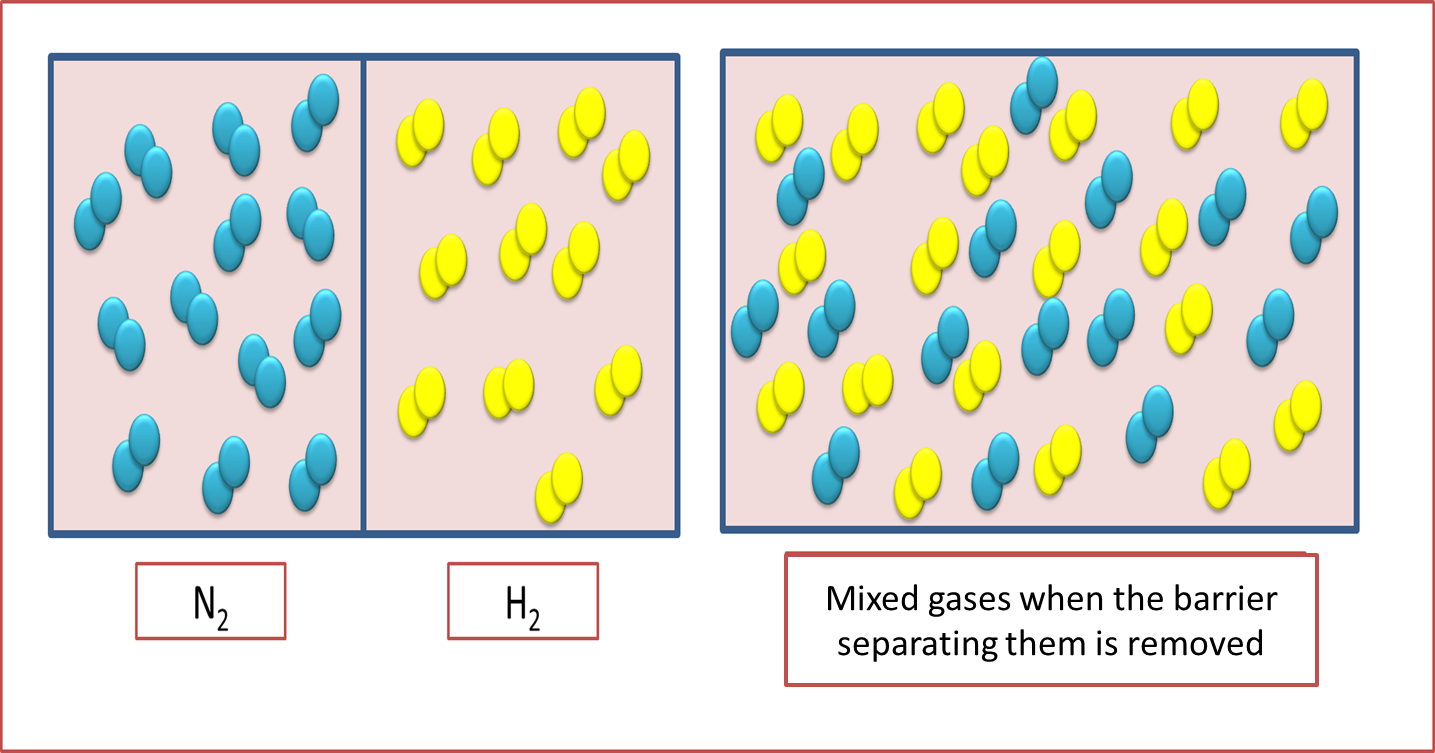
Fig. 4. Gas diffusion process. Author: @madridbg, through Power Point 2010.
On the other hand, effusion, is nothing more than the outflow of gas particles through a hole in the system. In this case, the outflow of gas is directly proportional to the velocity of the gas particles.
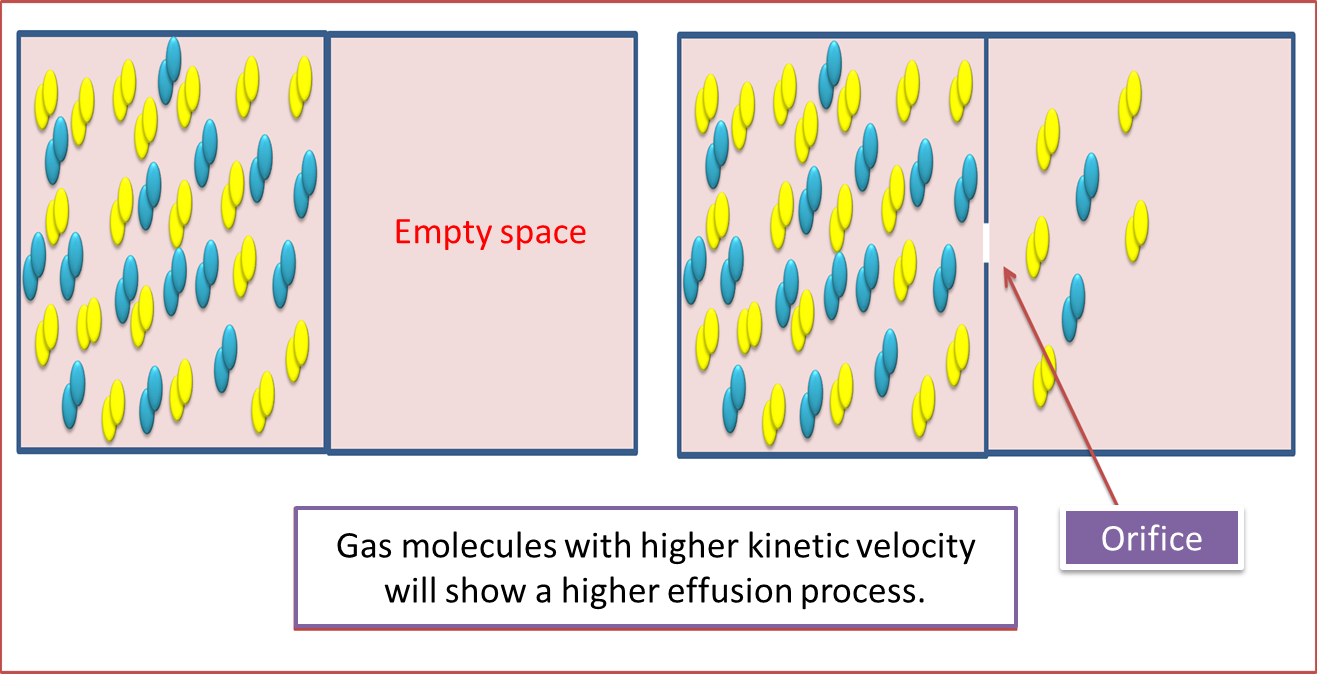
Fig. 5. Gas effusion process. Author: @madridbg, via Power Point 2010.
CHEMISTRY BEHIND AIR BAGS
The idea of writing this section is in function of correlating two aspects associated with the characteristics of science and the development of technology, on the one hand to implement the chemical fundamentals previously studied in the application and uses of air bags as a technological tool implemented in automobiles.
Therefore, the operation of this equipment integrates scientific knowledge associated with chemistry and engineering , on the one hand to generate the substance in exact quantities and concentrations and on the other to adapt it to the equipment in the most suitable way without causing major modifications e. the car.
In this sense, in this section of the subject we will only focus our attention on the scientific basis which is based on a set of chemical reactions that take place to produce the nitrogen gas (N2) needed in the airbags. Gas that meets certain safety parameters designed not to affect the life of the driver, among which are:
1. Non-toxic.
2. Because it is a cold gas it does not produce burns.
3. It must be generated in extremely short periods of time that allow its action at the moment of the collision.
4.
4. It must be stable for extended periods of time.
Under the above considerations, sodium azide (NaN3) is used, as a chemical substance that upon decomposition produces metallic sodium (Na) and gaseous nitrogen (N2), according to the following chemical equation.

Equation 2. Stoichiometric representation of nitrogen gas formation. Author: @madridbg, via Power Point 2010.
If we look at the equation, we realize that a heating triangle is present, which implies that for the process to be activated, it needs a small energy charge as the ignition point for the reaction to take place. In this sense the technological sensors are responsible for giving the answer for such a task.
Chemically in the above reaction, there is a serious drawback, the same lies in the generation of metallic sodium which is highly reactive when in contact with water and in small quantities produces large explosions due to these researchers in the field decided to prepare a mixture of molybdenum disulfide (MoS2), sulfur (S) and iron oxide and the corresponding sodium acid, which generates the expected nitrogen and allows to transform metallic sodium into non-reactive sodium disulfide.

Fig. 5. Representation of the operation of the air bags. Author: Orbea, (2017)
PRACTICAL CONTRIBUTION OF THE TOPIC
So far we made a conceptual tour of the chemical compounds necessary in the operation of airbags in automobiles, as well as the general fundamentals associated with the operation and dynamics of gases.
Therefore, I believe it is appropriate to instruct the reader in the correlation and integration of all the conceptual aspects addressed up to this point. For this task, we will quantitatively determine the amount of nitrogens produced from a given amount of sodium azide, a procedure that will allow us to implement the ideal gas laws and that we will carry out through a proposed exercise.
Proposed exercise:
The process of obtaining the gaseous nitrogen (N2) used for air bags, is carried out through the decomposition of sodium azide at high temperatures, applying the necessary correctives to transform the metallic sodium into a substance harmless to people. Based on the above, determine the amount of nitrogen obtained, using 70 grams of azide, a temperature of 299K or 26 °C and 735 mmHg of pressure. According to the following equation:

To answer the following question, we will be guided by the following set of steps:

Fig. 7. Step 1. to calculate the stoichiometry behind sodium azide . Author: @madridbg through Power Point 2010.
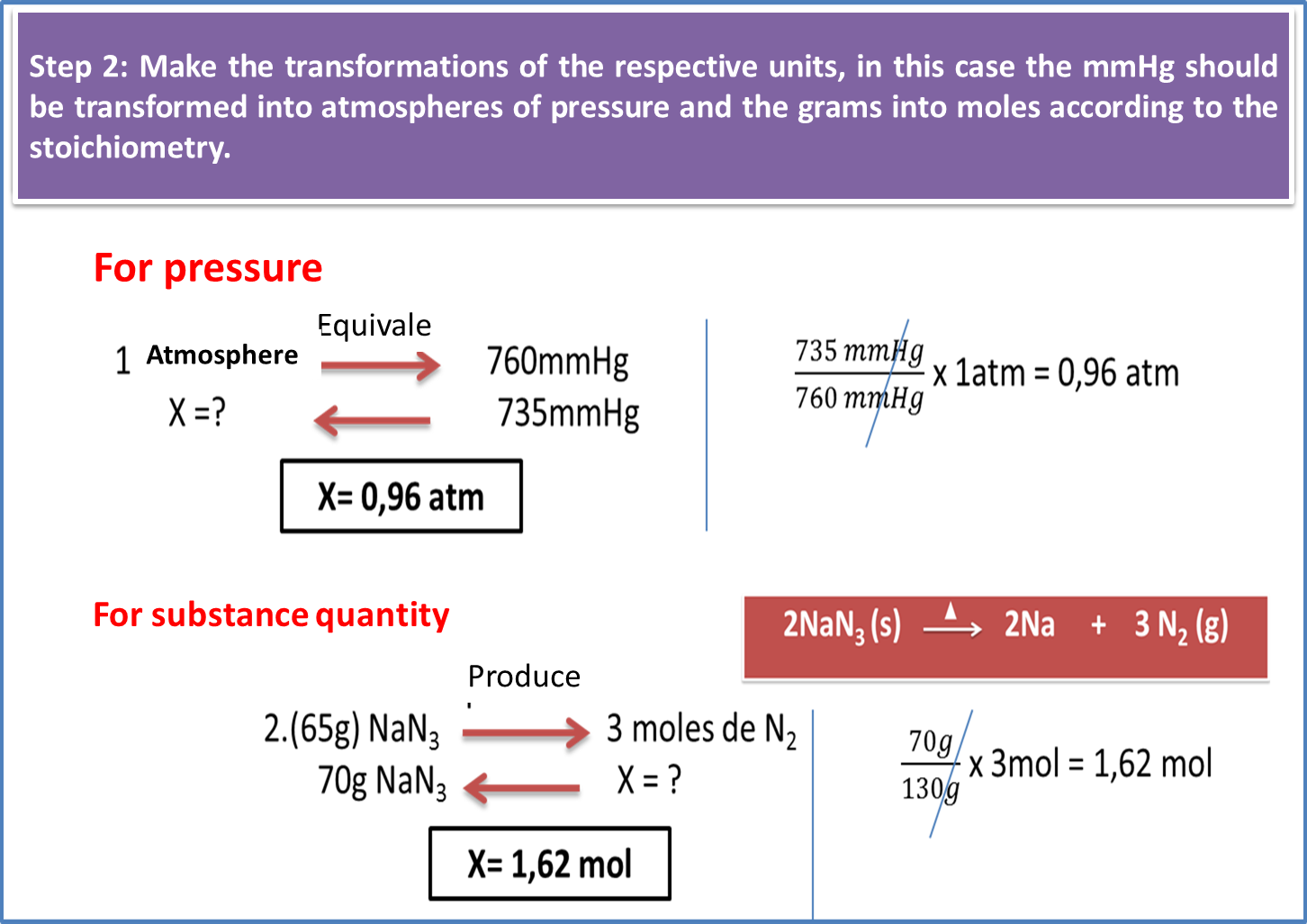
Fig. 8. Step 2 to calculate the stoichiometry behind sodium azide . Author: @madridbg through Power Point 2010.

Fig. 9. Step 3 to calculate the stoichiometry behind sodium azide . Author: @madridbg through Power Point 2010.
If we analyze the results obtained, we can see that a 70 g tablet of azide can be used as a raw material in the inflation of the bags used in airbags, since it produces a sufficient amount of nitrogen to inflate the device.
FINAL CONSIDERATIONS
As we progressed along the lines of this publication, we could observe that it allowed us to understand the applicative scopes of chemistry as a science, as well as its interaction with technological instruments, in the same way we could approach quantitatively measures implicit in the process, also the publication allowed us to approach the generalities of gases as well as the numerical scopes of the ideal gas law.
CONSULTED BIBLIOGRAPHY
[1] AZIDA DE SODIO N° CAS: 26628-22-8. Summary of Safety Data and Risk Factors in the Handling of Chemical Substances. UNIVERSITY OF SONORA, Division of Biological and Health Sciences. Institutional Environmental Health and Safety Program. PISSA –UNISON. Artículo: Acceso Online
[2] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[3] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[4] Orbea Hinojosa Luis Xavier. (2017). Design of an Airbag System Test Protocol through the Interpretation of Operating Oscillograms. Monthly magazine of the UIDE Guayaquil extension. INNOVA Research Journal, Vol 2, No. 9, 135-146.Artículo: Acceso Online
[5] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[6] WADE,LEROY. (2011). . QUÍMICA ORGÁNICA. VOLUMEN 2. SÉPTIMA EDICIÓN. PEARSON EDUCACIÓN, MÉXICO, 2011 ISBN: 978-607-32.()793•5. ÁREA: CIENCIAS
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/BGMadrid/status/1378457427654414344
#posh twitter:
https://twitter.com/BGMadrid/status/1378457427654414344?s=20
Interesting information, science is everywhere, it goes hand in hand with technology and it is something that must be very precise.
You couldn't have said it better @cryptoxicate, it is a fundamental union science and technology allows us to enjoy the advances we are having today. Thanks for your contribution
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.