AMORTIGUATING SOLUTIONS// Maintenance of acid-base balance at the metabolic level

Author: @madridbg, through Power Point 2010, using public domain images.
Welcome back to all those readers who are passionate about scientific content, associated with the chemical principles involved in our daily lives. Therefore, in this installment we will be addressing those concerning the maintenance process that occurs in our metabolism, specifically through our blood, giving a theoretical justification from the perspective of chemistry and its rationale.
As has been constant in my publications, this type of content is usually shared through the @stemsocial community who are trending in topics of scientific interest associated with chemistry, biology, mathematics, among others.
INTRODUCTION
A balanced life in the different areas of our existence is the most appropriate way to achieve a clear perspective and orientation in all the systems that compose us. Therefore, at the metabolic and homeostatic level this process is decisive in the preservation of life, which is why there are specialized substances that allow us to regulate the sudden changes that threaten the balance mentioned above.
Chemically, the substances responsible for maintaining the acid-base balance in organisms are known as buffering substances and are responsible for maintaining a dynamic balance in the pH of the system, because in our organisms products of catabolic and anabolic processes of biologically active molecules, large amounts of acids are produced that have to be controlled so as not to affect the naturally balanced system.
Buffering substances or buffer solutions are therefore responsible for the self-regulating process, of which we can mention the regulators bicarbonates, phosphates and hemoglobin. In this sense, through the publication we will address the chemical balances generated by these substances and we will focus on the maintenance that takes place in our blood, which allows the proper functioning of our system in general.
THE BRONSTED-LOWRY ACIDS AND BASES AND THEIR METABOLIC AFFECTIONS
In previous publications we mentioned the concepts of acid and bases proposed by BRONSTED-LOWRY, which established that any substance (ion or molecule) that has the ability to transfer a proton is considered an acid, while any substance that is able to accept a proton is defined as a base. [1].
Under these conceptions at the metabolic level there are a large number of substances that assume this behavior and that an uncontrolled production of them can generate a disorder in the level of acidity or basicity of our body, producing conditions such as acidosis, a disease generated as a result of excessive acid production or uncontrolled loss of bicarbonate in our body.
Similarly, this metabolic condition can originate due to an inefficient maintenance process in our blood system, allowing the accumulation of carbon dioxide in the blood, product of inconsistencies or conditions at the pulmonary level, where the concentration of oxygen becomes insufficient for the purification and maintenance of our blood.
On the other hand, the opposite effect can also occur where acidity levels are low and the basicity in the blood increases considerably, a condition known as alkalosis, where high amounts of alkaline or basic substances (bicarbonate) are produced and the amount of carbon dioxide is poor. Which is manifested by the presence of spasms, irritation and cramping.
This is why buffer systems are introduced as specialized systems to control the rise or fall of acidic or alkaline substances. Therefore, buffer solutions become a line of metabolic defense, among which we can highlight the regulators bicarbonates, phosphates and hemoglobin systems that we will study in depth in the following sections.
Fig. 2. Representation of red blood cells. Author: Vector8DIY
THE MEASUREMENT OF ACIDITY THROUGH HYDRO CONCENTRATION (pH)
The measure of acidity of a substance is determined by the concentration of hydronium ions (H3O+) in aqueous solution, operationally the pH can be determined or calculated by applying the method of Sorence, which states that pH is the negative logarithm of the concentration of hydronium ions in (mol/L). [3]
At the experimental level, the pH of a solution is measured with an apparatus called a peachimeter which is governed by a scale ranging from 1 to 14, with 7 being the neutral value, the numbers before this are set for acidic substances and the numbers after 7 are used for basic or alkaline substances.
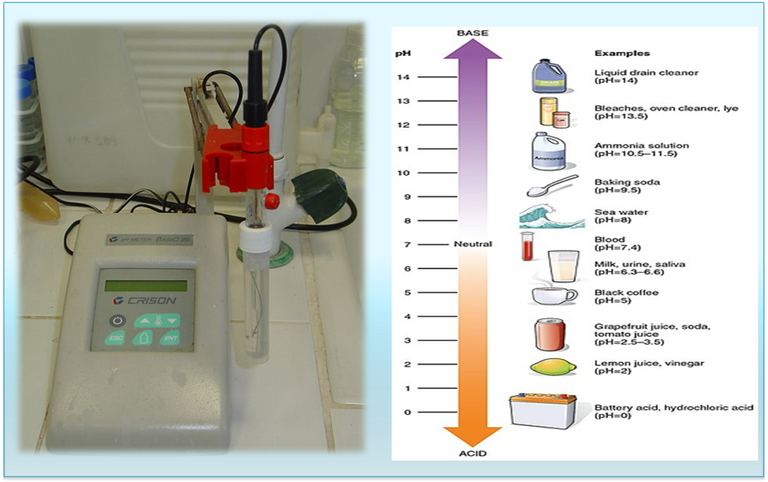
Fig. 3. pH indicators. Author: @madridbg, via Power Point 2010. Using public domain images
BEHAVIOR OF PHYSIOLOGICAL BUFFERING SYSTEMS IN THE MAINTENANCE OF BODY EQUILIBRIUM
Along the lines of this publication, we have made mention of a number of specialized compounds that are responsible for regulating our body's physiological systems. Therefore, we will follow a descriptive route that does not allow us to understand some of these systems and we will focus our attention on the set of chemical processes that take place at the metabolic level and that allow us to maintain a proper balance of the same.
Chemically and according to Chang. (2010), a buffer substance is a solution formed by a weak acid or a weak base and its conjugate salt, where both components must be present in the solution. These substances are very important for biological systems since enzymatic functioning and osmotic pressure balance is highly dependent on specific ranges in pH levels.
Fig. 4. Blood sampling. Author: pxfuel
Therefore, when talking about blood maintenance and purification, we must know that the vital fluid called blood is made up of a versatile set of chemical substances among which blood plasma and red blood cells (erythrocytes) stand out.
In relation to red blood cells, the major substances are hemoglobin and the enzyme carbonic anhydrase, which play a major role in the synthesis and degradation of the carbonic acid generated (H2CO3).
Consequently and by way of instructing the reader, in the functioning of this pair of compounds, we will address the interaction that occurs between the carbonate/carbonic acid buffers and hemoglobin, these being the main responsible for maintaining at an average value of 7.40 the pH levels.
In this sense, hemoglobin, chemically possesses several ionizable protons so it can be assumed as a weak acid that generates a conjugate base that allows it to behave as a buffer or buffering solution, according to the following equation.
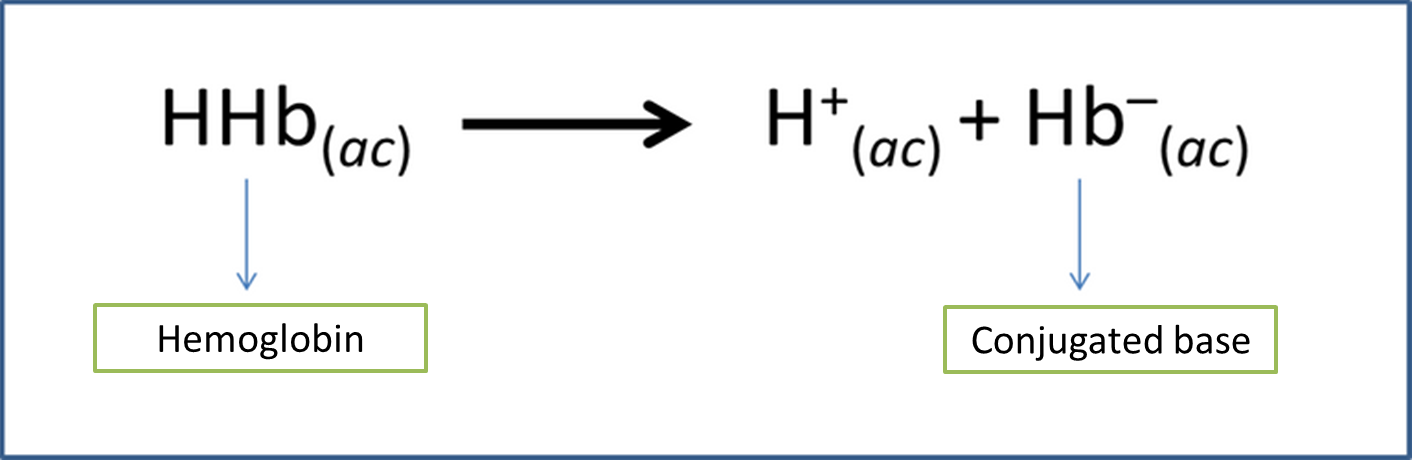
Equation 1. Hemoglobin conjugate pair. Author: @madridbg, via Power Point 2010.
The work of the buffer substances begins when a surplus of carbon dioxide is produced in the blood as a result of the metabolic processes of the organism, which is immediately transformed with the help of the enzyme carbonic anhydrase into carbonic acid, which is then ionized into bicarbonate and extracted from the bloodstream into the lungs so that the process is initiated with the help of hemoglobin. [2]

Equation 2. Carbonic acid formation. Author: @madridbg, via Power Point 2010. Adapted from McMURRY. (2008).
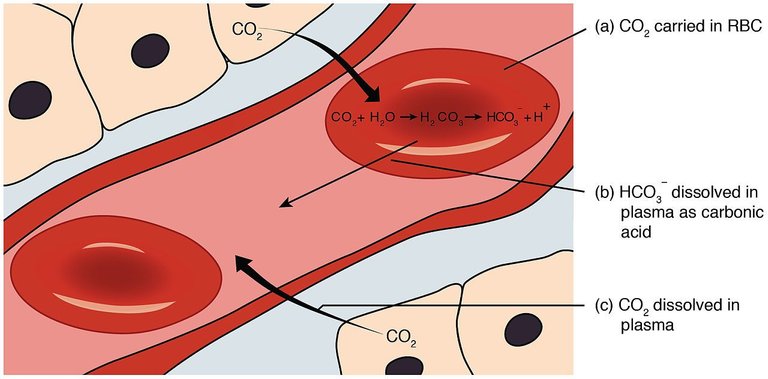
Fig. 5. Conjugated carbonic acid/bicarbonate pair. Author: OpenStax College. (2013).
In this sense, when blood with impurities returns to the lungs, bicarbonate molecules are activated, which binds with hemoglobin and allows the regeneration of ionized carbonic acid, a process that is constantly replicated and allows balance in the system.
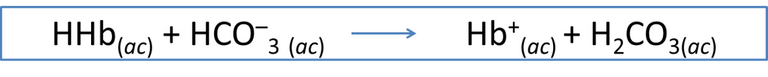
Equation 3. Hemoglobin and the generation of carbonic acid. Author: @madridbg, via Power Point 2010. Adapted from Chang. (2010).
CONTRIBUTIONS OF THE THEMATIC
Dear readers, if you have reached this point in the reading you have realized that we have addressed the importance of buffer solutions at the biological and metabolic level. In addition, we have chemically justified how pH variations are carried out in our organism, as well as the principles of self-regulation that our body follows to restore the values that are sometimes altered due to some inconsistency in our organs. This allows us to have a general understanding of the factors that affect our health.
BIBLIOGRAPHY CONSULTED
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] McMURRY E., John y Fay C., Robert. (2008). Química general. Quinta edición PEARSON EDUCACIÓN, México, 2009 ISBN: 978-970-26 1286-5.
[3] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[4] Túnez Fiñana Isaac, Galván Cejudo Aurora, Fernández Reyes Emilio. pH and buffers: Physiological buffers. Department of Biochemistry and Molecular Biology, School of Medicine. Córdoba.Artículo: Acceso Online
[5] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCATION, MEXICO, 2011 ISBN: 978-607-32.()793-5.
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/BGMadrid/status/1371663432253071362
#posh twitter:
https://twitter.com/BGMadrid/status/1371663432253071362?s=20
Congratulations @madridbg! You have completed the following achievement on the Hive blockchain and have been rewarded with new badge(s) :
Your next target is to reach 700 comments.
You can view your badges on your board and compare yourself to others in the Ranking
If you no longer want to receive notifications, reply to this comment with the word
STOPThanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.