ALKENES AND THEIR IMPACT ON VISION CHEMISTRY
Greetings and welcome dear readers, continuing with the scientific approach, this time we will be referring to the implications of certain hydrocarbons in the chemical processes associated with vision.
Specifically, we will be talking about alkenes, substances that we previously defined as hydrocarbons that are characterized by being constituted by carbon and hydrogen and by presenting double bonds in their structure.
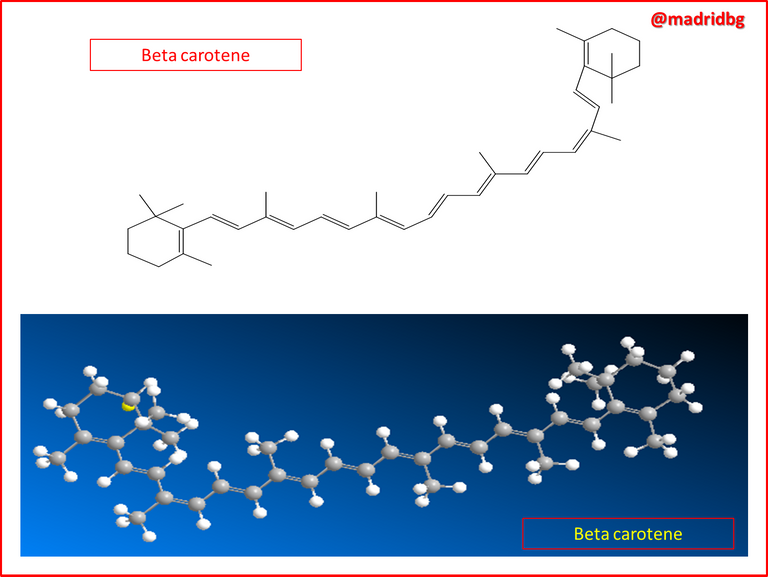
Among the most useful compounds within this organic family, we can get ethylene, which is associated with plant hormones, responsible for the ripening of fruits, also, we can identify the presence of the family of alkenes in the formation of beta carotene, whose compounds are essential in the coloration of various fruits and an example of them, we can see through the orange color of carrots which in turn provides large concentrations of vitamin A in our diet and exerts a direct action on some types of cancer.

Image courtesy of: Paul Diaconu
If we associate them with industrial processes, we realize that the simplest alkenes, referring to ethylene and propylene, are those that are produced in greater quantities at the industrial level, since their usefulness is diverse or multiple and is responsible for the synthesis or production of other materials, among which we can mention acetic acid, ethylene glycol, acetaldehyde, among other substances of great utility as solvents.
Another of the fundamental aspects regarding the usefulness of these organic compounds is due to the isomeric process presented by alkenes, understood as the spatial arrangement of the molecule, giving rise to Cis and Trans isomerism, which is of utmost importance in determining the usefulness of an organic molecule.
Consequently, this restricted rotation around the double bond of an alkene has generated great scientific interest since its configuration has direct chemical consequences that can be positive or negative. Let us recall the case of thalidomide where one of the active principles was used to relieve headaches and nausea while the other isomer was characterized by producing teratogenic effects in the fetuses of pregnant women.
Hence, the need to study and understand in depth the spatial organization of an organic compound, specifically alkenes. To exemplify this behavior let us analyze the following figure, note that the double bond has a restricted twist, that is, it cannot be easily mobilized so that within the compound 2-butene two different spatial arrangements can be presented that at the moment of isolating them as a compound we notice that they exert a different action one in relation to the other, compounds which we will call Cis isomers and Trans isomers.
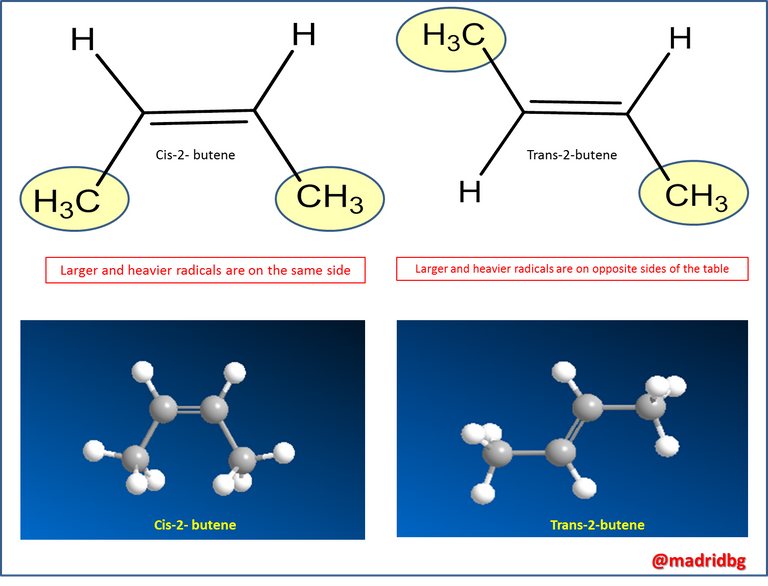
Now, to be able to establish which compound theoretically is of type Cis or otherwise Trans is enough to be guided by the radicals that are directly attached to the carbon atoms that share the double bond, so that when the radicals of greater size are in the same plane, We say that the compound is of type Cis or otherwise when they are in opposite planes we say that the isomers presented are of type Trans and as mentioned above this spatial arrangement provides benefits but can also exert negative actions, if we refer to the use in medicine or other substance of commercial interest.
In such a way that when approaching a study about the behavior of alkenes, it is necessary to understand their stability and although the interconversion of Cis or Trans type are not spontaneous, it can generate processes that hinder the synthesis of expected chemical compounds, hence we must maintain the degree of control over the specific experimental conditions, in which organic compounds belonging to the family of alkenes will be present.
For example, in the case of ethylene when it enters in the presence of a strong catalyst with acidic properties, we will notice that a greater reactivity is generated towards the Trans isomer representing an interconversion of more than 76% in comparison to its Cis isomer whose interconversion is only represented by 24%.
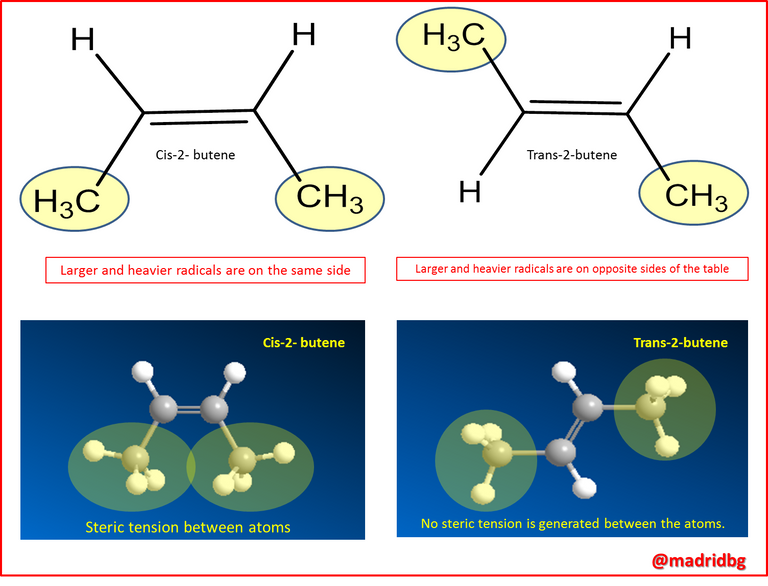
This allows us to predict that the Trans isomer is much more stable and reactive than the Cis isomers of 2-butene at least at room temperature, an action that is due to the steric or spatial tension that is generated between the two larger substituents when they are on the same side of a double bond, as we can see in the following figure.
In this sense, and from the above, let us analyze what is the incidence that alkenes exert on the chemistry of vision and for this it is necessary to understand that traditional medicine for many years has emphasized to divulge that the carrot is necessary for the care of our eyes, So that we are induced to consume a diet where the right dose of carrots is consumed, perhaps the relationship between carrots, alkenes and vision may seem absurd to many, but from scientific advances have been identified that the relationship is directly proportional to the variables that we are sharing.
In other words, the consumption of adequate concentrations of carrots has a direct and positive influence on people's vision and this is justified by the fact that carrots are rich in beta carotene and we previously explained that the characteristic orange color of carrots is due to the presence of groups or compounds belonging to the family of alkenes.
So at a chemical level, what occurs is a transformation process where the pre-established enzymes in the liver convert beta carotene into vitamin A, which has the property of oxidizing and producing an aldehyde called trans-retinal, this compound is able to enter into a process of isomerization around carbon 11 and 12 thus generating the cis-retinal isomer, which is a pigment sensitive to light and which is present in all visual systems of living organisms.
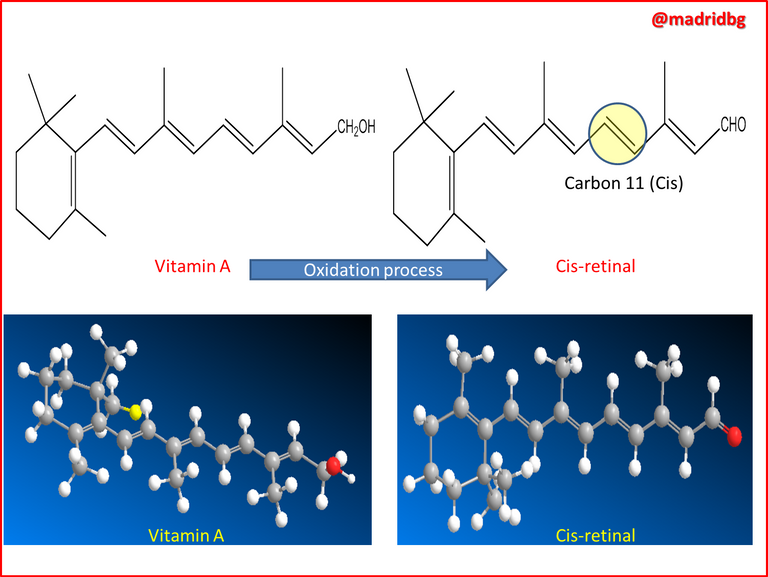
So that the understanding of the Cis - Trans isomerism of alkenes, allows us to understand what is the direct influence between the consumption of carrot and the improvement of our vision and as we have mentioned everything is executed through a spatial arrangement of the cis-retinal molecule, in the same way it is necessary to understand that there are two kinds of receptor cells that are sensitive to light in the retina of our eyes, which at a medical level are called rods and cones.
In this sense and at a medical level, it has been possible to identify more than 3 million rods that are responsible for perceiving through a dim light, while hundreds of millions of cones work to capture the bright light, as well as perceive all the characteristic colors and known so far.
Now, to understand a little more in depth about the chemical process it is necessary to know that when light reaches the rods in our eyes, the double bond generated between carbon 11 and carbon 12 of cis-retinal type, is isomerized and produces a compound called rhodopsin substance that is sensitive to light and consequently the process of cis - trans isomerization of rhodopsin is accompanied by a change in molecular geometry which in turn is responsible for generating the emission of nerve impulses to the brain.
Undoubtedly the isomerization process applied in the chemistry of vision is much more complex than what we have been able to describe here, however, it is necessary to understand that it has been through the advances in science and chemistry in general that we have been able to have a broader vision about the chemical reactions that are involved in the family of hydrocarbons associated with alkenes.
Therefore, I hope that this publication will be useful and we will continue to read each other in future issues.
BIBLIOGRAPHY CONSULTED
[1] Chang, R. (2010). Química. Decima edicion. McGraw-hill Interamericana editores. ISBN: 978-607-15-0307-7.
[2] Ralph, H. Petrucci, William S. Harwood, E. Geoffrey Herring. (2003). QUIMICA GENERAL. Octava edición. PEARSON EDUCACIÓN. S.A., Madrid.
[3] WADE,LEROY. (2011). . ORGANIC CHEMISTRY. VOLUME 2. SEVENTH EDITION. PEARSON EDUCATION, MEXICO, 2011 ISBN: 978-607-32.()793-5.
OF INTEREST
For more information related to the areas of science, technology, engineering and mathematics, do not hesitate to visit #stemsocial and #stem-espanol, they are communities that promote scientific advances in these areas

0
0
0.000
https://twitter.com/279290402/status/1618465600560590848
The rewards earned on this comment will go directly to the people( @madridbg ) sharing the post on Twitter as long as they are registered with @poshtoken. Sign up at https://hiveposh.com.
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider delegating to the @stemsocial account (85% of the curation rewards are returned).
You may also include @stemsocial as a beneficiary of the rewards of this post to get a stronger support.
Thank you for your support @stemsocial