HUMAN CIRCULATORY SYSTEM: The Blood As A Transport Medium
Blood is the fluid that flows through blood vessels, and the sight of blood is a sure sign that a vessel has ruptured. The loss of large amounts of blood has serious consequences: death will result if the ruptured vessels are not sealed and blood volume is not rapidly returned to normal. In an emergency, when there is no time to find out an accident victim’s blood group, he or she is given fluids called plasma expanders. These are isotonic fluids that contain no cells but which do increase blood volume.
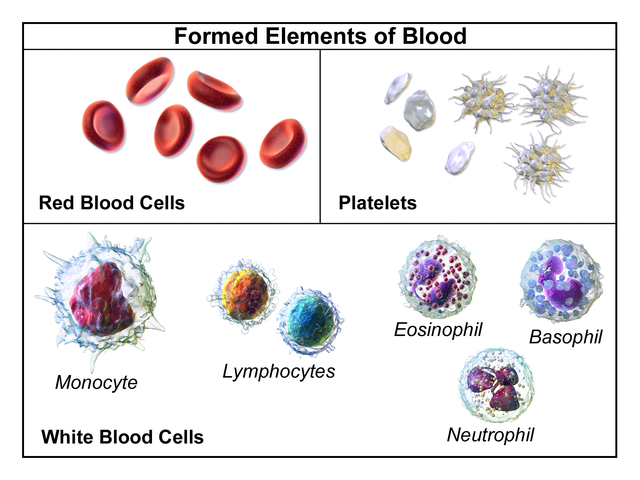
Blood is a complex mixture containing cells, cell fragments and a range of dissolved molecules. It does the following:
- Transports materials. Blood transports food and oxygen to respiring tissues. It also takes carbon dioxide and waste products away from respiring cells to the various organs that remove them. It carries hormones from endocrine glands to target organs.
- Distributes heat. The blood helps to maintain a constant body temperature by distributing heat from metabolically active organs, such as the liver and working muscles, to the rest of the body.
- Provides pressure. Many organs of the body depend on the physical pressure of the blood to carry out their function. For example, filtration in the kidney, formation of tissue fluid and erection of the penis all depend on blood pressure.
- Acts as a buffer. The blood contains many proteins and ions that act as buffers, keeping the pH constant by ‘mopping up’ any excess acid or alkali. Haemoglobin in red blood cells is an important buffer.
- Defends the body against infection.
WHAT’S IN BLOOD?
A centrifuged blood separates out into two distinct layers: a cellular portion called the haematocrit, and the plasma. The cellular portion contains red cells, white cells and platelets.
Plasma
Plasma is the fluid part of blood. The exact composition of the plasma varies greatly. For instance, in the hepatic portal vein, which leads from the intestines to the liver, the plasma is much richer in dissolved foods such as sugars, vitamins and amino acids than the plasma in a vein in another part of the body. The main constituents of plasma include the proteins (albumin antibodies, fibrinogen), salts (sodium, potassium, calcium chloride), products of digestion such as glucose, amino acids fatty acids, hormones (insuli, lipids, testosterone), heat, oxygen and waste products such as urea and carbon dioxide.
Red blood cells
They also known as erythrocytes, red cells are by far the most numerous type of cell in the blood. A single cubic millimetre of blood contains around 5 million, sometimes more. Red blood cells are made in the bone marrow of the vertebrae, ribs and pelvis by specialized stem cells. The turnover of red blood cells is very rapid: we make about 20 million per second! Red blood cells circulate for about 100 to 120 days before they are destroyed in the liver by the phagocytes called Kupffer cells and also in the spleen. Mammalian red blood cells have no nucleus, but, as well as haemoglobin, they contain the enzymes and chemicals that allow them to function effectively. Red blood cells have one function: they carry the respiratory gases. The haemoglobin (Hb) they contain picks up oxygen in the lungs and swaps it for carbon dioxide in the respiring tissues.
Having the Hb inside red blood cells rather than in solution in the plasma gives several advantages:
- A much greater volume of Hb can be carried in cells than could be dissolved in plasma.
- Hb can be kept in a favourable chemical environment to allow faster loading and unloading of respiratory gases.
- Hb molecules of a particular age are kept together and can be replaced easily when old.
- Hb in cells does not increase the solute potential of the blood (free Hb would).
Red blood cells have a very regular shape, described as a biconcave disc. This shape is maintained by the cytoskeleton, a complex but flexible internal scaffolding made from protein fibres. When you think about what red blood cells do, it is easy to see why this is a good shape for them. They must have a large enough volume to carry useful amounts of oxygen but they also need a large enough surface area to load and unload it quickly. The biconcave shape is an efficient compromise between the maximum volume of a sphere and the maximum surface area of a flat disc.
Red blood cells are distorted when they have to squeeze through capillaries that often have a lumen slightly smaller than the diameter of the cells. Red blood cells rarely cause blockages because they are flexible and have a smooth surface.
BLOOD AS A TRANSPORT MEDIUM
Oxygen is carried in the blood in two ways: 98 per cent travels as oxyhaemoglobin, the oxygen-haemoglobin complex; the remaining 2 per cent is dissolved in the plasma. Carbon dioxide is carried in three ways as HCO3- ions in the plasma (70 per cent), as carboxyhaemoglobin (23 per cent) and in simple solution in the plasma (7 per cent).
The structure of haemoglobin
The haemoglobin molecule is a conjugated protein with a relative molecular mass of about 64 500 kilodaltons. Conjugated means ‘joined’. A conjugated protein, such as haemoglobin, consists of a protein attached to a prosthetic group (a non-protein). The prosthetic group in haemoglobin contains iron, so we need a regular supply of iron in our diet to make this vital chemical. Each Hb molecule consists of four globin sub-units consisting of a polypeptide chain and a prosthetic group called haem. At the centre of each haem is an iron ion (Fe2+), which combines with oxygen. There are four haem groups, so the overall equation for the reaction is:
Hb + 4O2 → HbO3
Haemoglobin + oxygen → oxyhaemoglobin
The polypeptide chains hold the haem groups in place and help to load oxygen. When the first haem group combines with an oxygen molecule, the haemoglobin molecule alters its shape so that the next haem group is exposed, making the loading of the subsequent oxygen molecules easier. Conjugated means ‘joined’. A conjugated protein, such as haemoglobin, consists of a protein attached to a prosthetic group (a non-protein). The prosthetic group in haemoglobin contains iron, so we need a regular supply of iron in our diet to make this vital chemical.
Haemoglobin in action
There are many substances that react readily with oxygen, but haemoglobin is one of the few that can combine with oxygen where it is abundant and then release it when the concentration falls. The concentration of dissolved oxygen is often referred to as the partial pressure, or tension. The greater the concentration of dissolved oxygen, the higher the partial pressure or tension. This property is illustrated in a graph called an oxygen dissociation curve. This is plotted by analyzing the percentage of Hb saturated with oxygen at different concentrations of oxygen. The graph shows that Hb becomes fully saturated with oxygen at the concentrations of oxygen found in the lungs, and gives up a relatively large proportion of its oxygen in the lower oxygen concentrations that occur in the tissues.
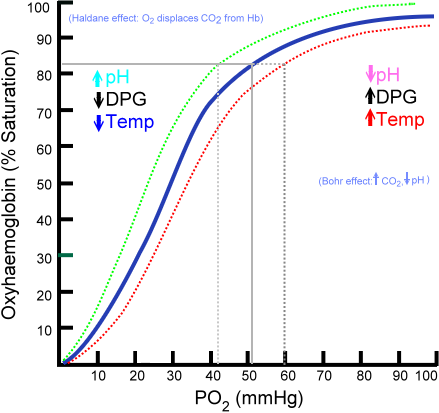
The dissociation curve was plotted using mixtures of gases in which the concentration of carbon dioxide was constant. It shows that, at higher concentration of carbon dioxide, the curve moves to the right. This is a vital point: it shows that carbon dioxide lowers the affinity of Hb for oxygen. This means that when carbon dioxide concentration is higher, Hb does not hold on to its oxygen quite as well: Hb therefore tends to give up oxygen in areas of high carbon dioxide – such as in the respiring tissues that need it most. This lowering of affinity by carbon dioxide is called the Bohr effect, or the Bohr shift.
Fetal haemoglobin
Before birth, a mammalian fetus must obtain oxygen from its mother via the placenta. If fetal haemoglobin had the same affinity for oxygen as the mother’s haemoglobin, no transfer of oxygen to the fetus would be possible. Fetal haemoglobin has a higher affinity for oxygen than adult Hb. It can therefore pick up oxygen in the same conditions that cause the maternal blood to release it. After birth, the baby’s body makes adult Hb, which gradually replaces the fetal version.
HAEMOGLOBIN IN OTHER ORGANISMS
Many invertebrates make use of haemoglobin to survive in oxygen-poor waters. Tubifex (sludge) worms, bloodworms (the larvae of the chironomid midge) and some ramshorn snails (of the genus Planorbis) all owe their red colour to their haemoglobin. These species are often used as indicators of polluted water. The oxygen dissociation curve of an organism like Tubifex that thrives in low-oxygen conditions is well to the left of normal human haemoglobin.
Another invertebrate that makes good use of haemoglobin is the giant tube worm Riftia pachyptia that lives in deep sea volcanic vents. This grows to a massive size, well over 2 metres, and survives by means of its population of symbiotic bacteria that hydrogen sulfide to make its food. The worm has a deep red plume at one end that is filled with haemoglobin complexes. These absorb oxygen, carbon dioxide and hydrogen sulfide from the water. These tube-worm haemoglobins are remarkable as they can carry oxygen in the presence of sulfide.

UNLOADING OXYGEN AT THE TISSUES
The series of events that lead to Hb unloading its oxygen in the respiring tissues is highlighted below:
Step 1: Carbon dioxide diffuses from the respiring tissue, through the capillary wall and the plasma, into the red blood cell.
Step 2: Inside the red cell, the enzyme carbonic anhydrase catalyzes the conversion of carbon dioxide into carbonic acid (H2CO3).
Step 3: The hydrogen ions released by the carbonic acid make the haemoglobin molecules less stable, causing them to release the four oxygen molecules.
Step 4: The oxygen is free to diffuse into the respiring tissues.
Step 5: The accumulating hydrogencarbonate ions diffuse out into the plasma, leaving the inside of the red cell with a net positive charge.
Step 6: To maintain a neutral charge, chloride ions (the commonest negative ions in plasma) diffuse into the cell; this is called the chloride shift
Myoglobin and muscles
Mammalian muscle contains the respiratory pigment myoglobin (myo = muscle). The myoglobin molecule is one-quarter of an Hb molecule: it consists of one polypeptide chain and one haem, and combines with one oxygen molecule. The presence of myoglobin in muscles is associated with endurance. Chickens, not known for their flying ability, have flight muscles that contain little myoglobin: this is why chicken breast meat is pale. In contrast, ducks and geese are accomplished long-distance fliers, and have dark breast meat because of the high levels of myoglobin.
Myoglobin has a higher affinity for oxygen than haemoglobin and so it can pick up oxygen from haemoglobin and store it until the muscle becomes short of oxygen. When this happens (during exercise, for example), the oxygen tension in the muscle drops. Hb has no more oxygen to deliver and myoglobin then releases its oxygen.
Haemoglobin across the animal kingdom
Animals that live at high altitudes, where there is a low partial pressure of oxygen, often have haemoglobin that has a higher affinity for oxygen than usual. In South America for example, llamas, alpacas and vicunas have an oxygen dissociation curve which is well to the left of that for animals that live closer to sea level.
A lot of aquatic invertebrates also make use of haemoglobin. There is much less oxygen in water than in air, and the warmer the water is, the less oxygen it can hold. In polluted water, oxygen can be especially scarce because the bacteria compete for what little there is. Several species all owe their red coloration to the presence of haemoglobin:
- Tubifex worms live in poorly oxygenated mud at the bottom of rivers and ponds. They burrow head first into the mud, leaving their tail sticking out.
- Chironomid midge larvae (bloodworms) live in burrows, also in poorly oxygenated mud. They must come out from time to time to replenish their oxygen stores. The haemoglobin in their tissues allows them to store oxygen and stay in the burrow for longer, thus avoiding predators.
Thank you for reading.
REFERENCES
- Blood
- Blood As A Transport Medium
- Plasma
- Blood- Britannica
- Transport system
- BLOOD AS A TRANSPORT MEDIUM
- Hemoglobin and its measurement
- Haemoglobin-Wikipedia
- Animals science
- oxygen transport
- oxygen transport
- Gas transport oxygen and hemoglobin
- myoglobin
- Myoglobin
- Myoglobin and muscles
- haemoglobin determinant
This post was shared and voted inside the discord by the curators team of discovery-it
Join our community! hive-193212
Discovery-it is also a Witness, vote for us here
Delegate to us for passive income. Check our 80% fee-back Program
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Thanks for using the STEMsocial app, which gives you stronger support. Including @stemsocial as a beneficiary could yield even more support next time.