pH changes during NaCl electrolysis
As we know, electrolysis is a process by which a chemical reaction takes place when electric current is applied to a solution, causing oxide-reduction reactions to occur on both electrodes. And during the process there are changes in pH in the vicinity of the electrodes that are imperceptible to the eye, but with an appropriate pH indicator, can be shown by interesting color changes around both electrodes.

Source: @emiliomoron.
Well, this time we will test this phenomenon by means of a simple experiment that we can do at home. In the following video we can check the pH changes that occur in a sodium chloride (NaCl) solution during electrolysis.
Source: @emiliomoron.
As you have seen, using the indicator makes it easy to identify the pH changes that occur around the electrodes. Let's analyze the situation a little more.
The following picture shows what happens at the cathode during the electrolysis of the aqueous NaCl solution, which is dissociated into Na+ and Cl-.
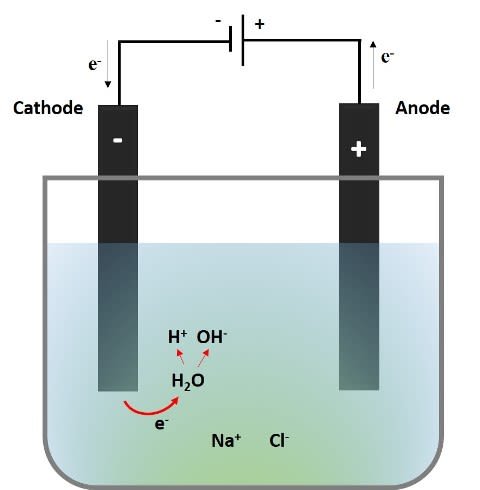
Source: @emiliomoron.
When the circuit is established the sodium ions (Na+) will be mobilized towards the cathode, but they do not suffer any reaction since the reduction potential of the hydrogen reduction reaction of water is higher, and this is the reaction that is carried out in preference.
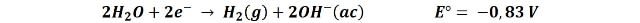
What we observe in the video is that an electron is transferred from the cathode to a water molecule releasing an atomic hydrogen, and with the hydrogen released from another water molecule, are joined to form a gaseous hydrogen molecule (the bubbles we observe) and the hydroxyl ions (OH-) that give the basic character to the solution, so that in the area near the cathode the pH increases, and the indicator turns from purple to green.
In this experiment I am using as pH indicator the purple cabbage extract, this contains natural pigments that give that purple color, but in basic medium they change to green color and in acid medium they become red color.
On the anode, however, the process may be a little more complex, as there are two competing reactions.
1- The oxidation of the oxygen in the water to form gaseous oxygen:
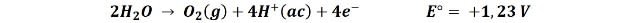
2- The oxidation of the chloride ion to form chlorine gas:
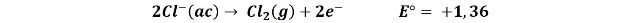
That the first or second reaction occurs is somewhat difficult to predict since the value of the reduction potential of both reactions is very close. However we can presume that diluted solutions will lead to the first reaction, releasing oxygen and in concentrated solutions the tendency is for chlorine to be produced.
Then, considering that we are working with a diluted solution of NaCl; in the following image the process that occurs at the anode is represented.
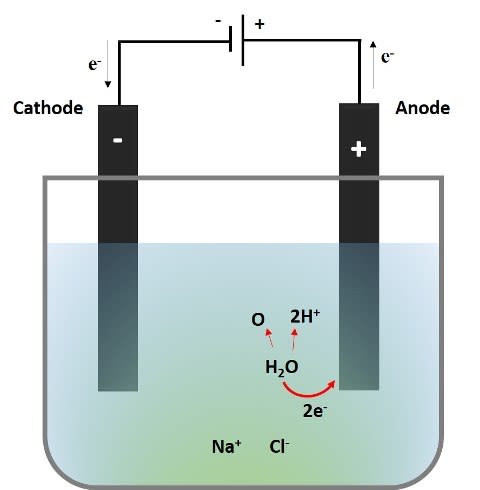
Source: @emiliomoron
What we see is that two electrons from the oxygen in the water are transferred to the anode, the monoatomic oxygen will combine with another monoatomic oxygen and be released as a gas, while the hydronium ions (H+) released give the acidic character to the solution near the anode, thus changing the color of the indicator from purple to red.
Well friends, this type of material allows us to visualize clearly the processes of oxidation-reduction that take place during the electrolysis of a NaCl solution, and can be used by teachers and students as reference material and educational support, or as a guide to carry out the reaction at home or in the classroom, as it requires very few materials and all are very easy to achieve, in addition, that very visible results are achieved that help to fix the knowledge taught in the classroom.
I hope you liked this simple but interesting experiment. See you next time.
References:
Wikipedia.com. Electrolysis.
Wikipedia.com. Table of reduction potentials.
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Thanks for using the STEMsocial app and including @stemsocial as a beneficiary, which give you stronger support.