Iron oxidation process. With demonstration
As we know, some metals, especially iron, suffer an oxidation reaction when exposed to the environment, forming what we know as rust.

Rust appearance. Source: pixabay.com.
The economic impact this process has on our current technology is considerable, as every year we spend significant amounts of money to prevent our iron elements and structures from rusting and eventually suffering the detrimental effects of corrosion. But the oxidation of iron not only has an economic impact, but also a visual one, because when rust is formed, Fe2O3, no doubt we all have an ugly perception of that reddish coat that covers the surface of iron objects, even we are disgusted to touch the rusted objects and to stain our hands or clothes with that reddish compound, that is why we are so concerned about keeping the iron objects well painted.
But why does this rust form?
The simple answer is that iron suffers an oxidation-reduction reaction when exposed to the environment. And with this post I want to offer a simple explanation of the reactions involved in the process.
First we must understand that oxidation is a chemical reaction where a species suffers the loss of electrons[1].
But as we may have already noticed, iron oxidation is very fast when compared to other metals, such as copper or silver, not to mention those that do not corrode and always look shiny, such as gold and platinum.
To begin with we must say that with few exceptions, the oxidation of metals is inevitable, this is mainly because in nature they are not found in a pure state but rather combined with other elements, forming oxides, carbonates or sulfates. When we produce steel, large amounts of energy are used to refine the iron and separate it from the rest of the elements with which it is combined, so its pure metal state is not its most stable form, so we could see oxidation as a spontaneous process used to return the metal to its natural state. In the case of iron its most common natural form is hematite, which is an oxide of formula Fe2O3 [2].
Now, what does this process depend on, and why does it occur so easily in iron?
If we have reviewed a little the theory of oxidation-reduction reactions, we can establish that the oxidation of metals depends on their standard reduction potentials. In the following picture we can see the standard reduction potentials for some compounds.
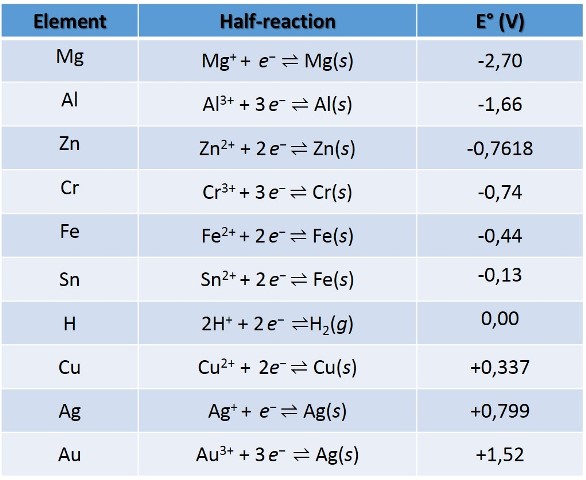
Source: data taken from wikipedia.com
As we can see, for the pair Fe2+/Fe the reduction potential is low, -0.44 V, if we compare it with the pair Au3+/Au, this indicates that the tendency of metallic iron is to oxidize when it is in contact with other elements that have a higher standard reduction potential, and that the gold cation acts basically as an oxidant, almost as good as chlorine. Therefore, iron oxidizes easily while gold is not allowed to oxidize.
And this is due to the electronic configuration that each metal has. The electronic configuration of iron does not favor its resistance to corrosion, since it does not allow it to protect its electrons and prevent them from being torn out, so that in contact with water and oxygen it oxidizes, that is, it suffers the loss of electrons.
Basically the process of iron oxidation can be described in two stages.
Stages of iron oxidation
Oxidation of metallic iron to iron (II)
In this first stage, a process analogous to what happens in galvanic piles occurs. On the surface of the metal some zones are produced that function as small electrochemical cells, on the one hand, in some zones the oxidation reaction takes place (anodic zones) and in other zones the reduction reaction (cathode zones).
In the anodic areas, iron oxidation occurs according to the semi-reaction:
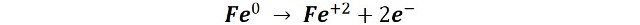
And in the cathodic zones the reduction of atmospheric oxygen is produced:
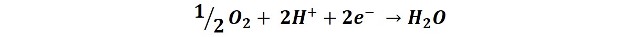
The global reaction can be represented in the form:
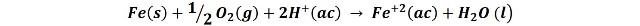
If we evaluate the potential of the reaction in this galvanic cell, calculated from the difference of potentials of each semi-reaction, we have left:
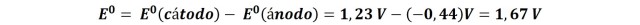
Which is a considerably high potential, let us remember that a positive potential difference in a pile implies a negative Gibbs free energy variation, that is to say, it is a reaction that occurs spontaneously.
Iron (II) to Iron (III) oxidation
In this stage, iron oxide Fe2O3 is produced. This iron (III) oxide is what precipitates forming rust on the surface of the metal, and is what we know when the iron is oxidized, when we observe it with that reddish or orange color.

And to think that one day it was a shiny screw. Source: @emiliomoron.
In this second stage the process continues according to the following reaction:
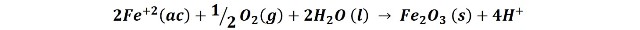
We see in the reaction that the H+ ions are also regenerated, which makes it easier for the process to continue.
The iron (III) oxide is deposited on the surface of the iron object in the form of that fine reddish-brown powder, forming an opaque and porous layer that does not fix strongly to the surface, let's remember that that is why we hate to stain our hands when we touch a rusty object. That's why that layer of rust comes off easily, exposing the surface of the metal and allowing the process to continue to the deeper layers.

Rust stains on the fingers. Source: @emiliomoron.
Over time, an entire iron object will become covered with rust. This is natural, since, as we have said, iron is a refined metal and is only returning to its natural state.
In the following experiment we can visualize how iron oxidizes.
Demonstration
We will take a disposable plate and add some water with dissolved salt, and in it we will place an iron nail (the water in each plate should be enough to leave part of the surface of the nail exposed).

Start of the experiment. Source: @emiliomoron.
After a few hours we begin to observe that the water in the proximity of the nail is turning orange, which gives us indications that the oxidation reaction has begun.

Appearance of water in the proximity of the nail. Source: @emiliomoron.
As we have said, this reaction is carried out because in the nail some zones are produced that work as small electrochemical cells, on the one side, in some zones they work as cathode and others as anode.
The anodic and cathode zones are not necessarily located in opposite places, usually the cathode zones are usually located where the oxygen concentration is higher, and conversely, the anodic zones are usually located where the oxygen concentration is lower, this is common in the environment as the surface of the metal exposed to the atmosphere can vary, for example; what happens when we have part of the nail submerged in the water, as the oxygen concentration is higher in the surface exposed to the air in this tendency to locate the cathode zone, not being observed as much oxidation in this part. And the anodic zones will be preferably located in the submerged surface, with less oxygen concentration, producing the oxidation of the nail in this part, dissolving the iron ions in the solution.
The iron Fe+2 ions react with the dissolved oxygen in the water to produce an orange-red hydrated ferric oxide precipitate.
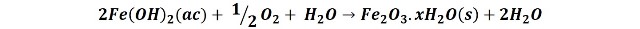
Therefore, with the passage of time the nail oxidizes into ferric oxide. In the following images we can compare how the solution is colored with the ferric oxide as the time of the experiment passes.
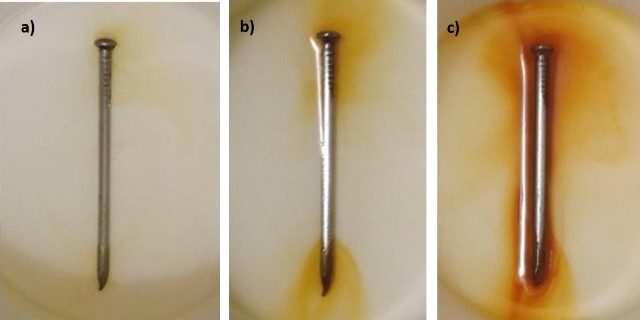
Comparison of oxidation progress. a) 4 hours, b) 12 hours and c) 24 hours. Source: @emiliomoron.
As oxidation is an electrochemical process, which requires the transfer of load; the dissolved salt increases the conductivity of the medium favoring the transport of load in the solution, and accelerates the process. This is why in saline environments the corrosion of iron is even faster.
In this image we can see the water collected from the plate, the orange colloidal-looking precipitate is hydrated ferric oxide, the same that has a reddish color on the iron object.

Appearance of the solution containing the ferric oxide. Source: @emiliomoron.
Well friends, with this article I hope to have shared information of your interest about the oxidation process. As we have seen, the oxidation of an iron element exposed to humidity and air does not take long to be noticed, making it easy to demonstrate with this simple experiment, so that the oxidation-reduction reactions involved in the process can be easily exposed.
Thank you very much for reading the post. See you next time!
Reference
- Chang, R. (2002). Chemistry. 7ma, McGraw-Hill.
- Wikipedia.com. Hematite.
Interesting piece, especially with the demonstration.
Thank you very much @gentleshaid. Thank you for appreciating it.
You have written well and i enjoyed the details put into explaining the rusting processs. Good job @emiliomoron
Thank you very much @mengene, I am glad I wrote it well and that the process involved in oxidation was understood in a simple form. Thank you for coming to read.
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Thanks for using the STEMsocial app and including @stemsocial as a beneficiary, which give you stronger support.
Thank you very much for your support friends!
Good job! I wish my chemistry lessons would be as interesting as your post lol!
Thanks friend, I'm glad you found it interesting, that's the idea of sharing educational articles in chemistry, that are easy to understand. Thank you very much for leaving your comment.
@rot-trip has a lot of rust and it does not like being rusty :P
I don't think anyone likes it haha. Thanks for coming by and reading!