Carbohydrate Chemistry
Carbohydrates are the most abundant biomolecules in nature and, together with proteins and fats, are the main nutrients found in food, mainly of plant origin and are our main source of energy. But these molecules comprise a vast field of carbon chemistry, so today I present important information about this type of compounds that will allow us to understand a little about the properties of these molecules and their nutritional value.

Carbohydrates are our principal source of energy. Source: edited image, original from pixinio.com, CC0.
Understanding the chemistry of dietary carbohydrates is very important for chemistry professionals who research and control the manufacture and properties of foods, since the chemistry of these compounds could vary during their preparation in the processing facility, during their storage or even during their final preparation in our kitchen. But having a notion of this group of foods is also very important for us as consumers, in fact, many think that they should eliminate them from the diet because they are fattening, but they are very necessary for the functioning and balance of our body, besides, this group of foods is so large that they would be very difficult to eliminate completely, or they think that only bread is a carbohydrate!
To begin with we must know that in our diet we have included a large number of carbohydrates, from simple, such as the so-called monosaccharides, to complex, such as polysaccharides. That is why we must understand how their structures determine their properties, and in turn, how their properties determine their uses. So let's make a small description of this important food group in this post.
Carbohydrates in the diet
First of all, we must say that carbohydrates are a type of molecule constituted by carbon, hydrogen and oxygen, which generally have a 2:1 ratio of hydrogen-oxygen atoms, so they conform to the empirical formula CmH2On. In general terms, they constitute a group of molecules known as saccharides, which include sugars, starch and cellulose[1].

Common sugar is one of the most widely consumed saccharides. Source: @emiliomoron.
Carbohydrates in food are mainly sugars and sugar derivatives, some of the most common being sucrose or common cane sugar (C12H22O11), glucose (C6H12O6) and fructose (C6H12O6).
Although in the food area, the term carbohydrate is commonly and informally associated with any food rich in sugar, such as sweets and desserts, or with the complex carbohydrate starch, such as bread and pasta.
But a better way to classify this group of carbon compounds is according to their degree of polymerization, being divided mainly into three groups, these being sugars, oligosaccharides and polysaccharides, as shown in the following table.
Table 1. Carbohydrates present in our diet

Data taken from Wikipedia.com.
Monosaccharides and disaccharides
Monosaccharides are the simplest carbohydrates, i.e., they cannot be hydrolyzed to smaller hydrates, being aldehydes and ketones with two or more hydroxyl groups that comply with the formula (CH2O)n.
On the other side, when two monosaccharides are joined by covalent bonds they form a disaccharide, which represents the simplest form of polysaccharide.
Monosaccharides and disaccharides are constantly present in our diet, particularly in sweet fruits and vegetables, and have been used to add sweetness to foods.
The most common monosaccharides are three:
- Glucose: is the sugar we can find most abundantly in nature, it is found in free form in many fruits, and is categorized as a hexose since it contains 6 carbon atoms in its structure, C6H12O6. In general, almost any other carbohydrate we ingest ends up being converted into glucose through different biochemical reactions, as this is the main source of energy for our cells.
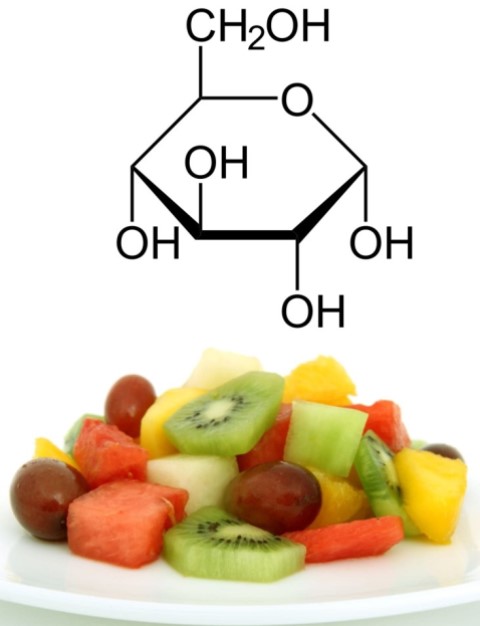
Structure of glucose. Source: edited image, contains public domain images: structure and fruits.
- Galactose: this simple sugar is also a hexose and is part of the glycolipids and glycoproteins present in cells. It is also considered an aldohexose because it contains an aldehyde group at its end and is a C-4 epimer of glucose, i.e. it has the same molecular formula as glucose but differs in the orientation of the alcohol group attached to carbon number 4. A glucose molecule attached to a galactose molecule forms lactose, so the main source of galactose comes from milk intake[2].
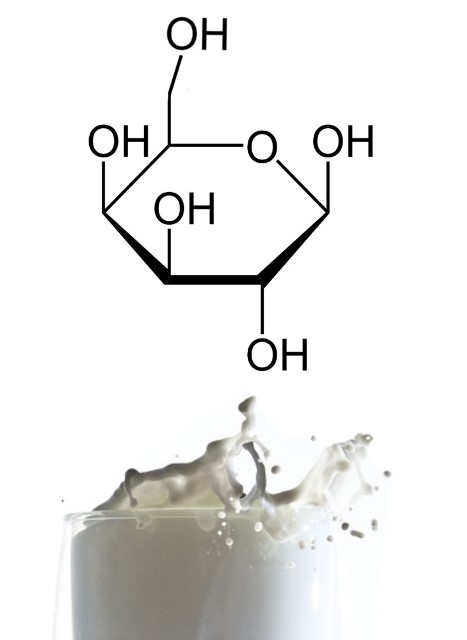
Structure of galactose. Source: edited image, contains public domain images: structure and milk.
- Fructose: is a monosaccharide very abundant in sweet fruits and honey. It is also an isomer of glucose, so they have the same molecular formula but with different structure; this compound differs in the presence of a ketone group in the chemical structure, so it is also said to be a ketohexose.
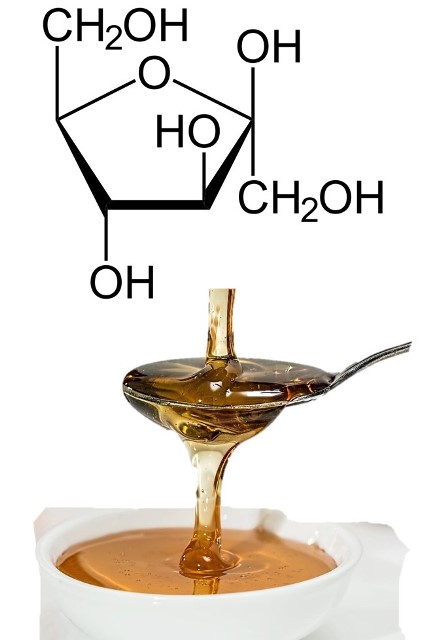
Structure of fructose. Source: edited image, contains public domain images: structure and honey.
And among the most common disaccharides we can name:
- Sucrose: better known as common sugar, it is perhaps the most consumed carbohydrate in the world, it is mostly obtained from sugar cane and sugar beet. This carbohydrate is a disaccharide composed by the union of a glucose molecule and a fructose molecule.
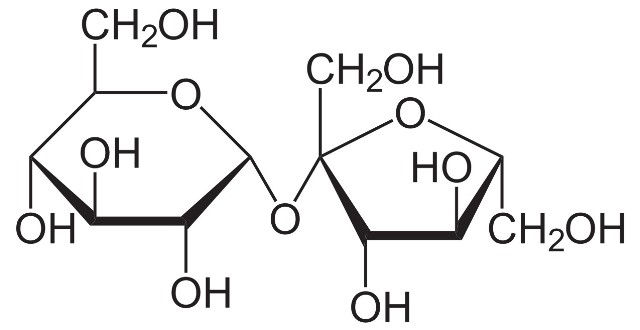
Structural formula of sucrose. Source: Wikipedia.com.
- Lactose: it is known as the milk sugar, because it is naturally synthesized in the mammary tissue of women, cows and females of other mammals. This disaccharide is formed by the union of a glucose molecule and a galactose molecule[3]. Lactose is obtained commercially from cow's milk whey, obtained as a by-product of cheese production, and is used in the preparation of infant food supplements.
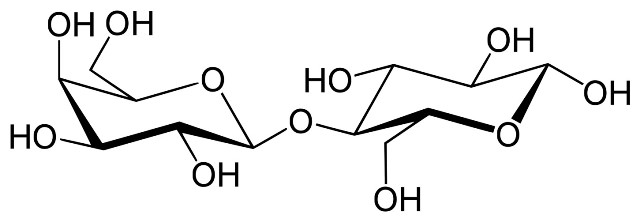
Structural formula of beta-lactose. Source: Wikipedia.com.
- Maltose: this disaccharide is formed by the union of two glucose molecules, and is produced in low percentages in sprouted grains, which is why it appears in products such as beer[4]. During brewing, maltose is produced by the action of germinating barley, or malt, on starch, and is therefore known as malt sugar.
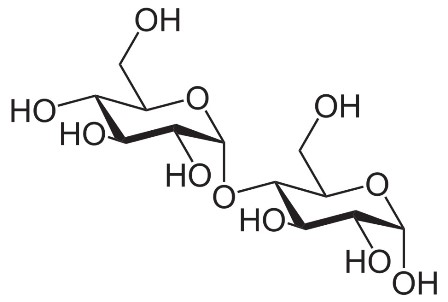
Structural formula of alpha-maltose. Source: Wikipedia.com.
As we can see, these three disaccharides contain a glucose molecule, which is linked to another monosaccharide by an alpha glycosidic bond, which is represented in their structures in the form of a "V", the term glycosidic refers to links between sugars. And if we look at lactose, this is different, since it contains a beta bond, which requires a special enzyme to break this bond, and the absence of activity of this enzyme is what makes people suffer from a condition called lactose intolerance.

General form of the three most common disaccharides. Source: @emiliomoron.
Oligosaccharides and polysaccharides
These are the most complex carbohydrates. Oligosaccharides are part of the fiber of plant tissue, and are molecules that are constituted by the union of several monosaccharides, usually from 2 to 10, although the most complex ones can have even 20; and from three onwards they can be linear or branched by glycosidic bonds[4]. The simplest group of oligosaccharides are the disaccharides just mentioned, while other more complex ones have important properties for health, such as the fructooligosaccharides present in many fruits and vegetables such as onions, garlic, asparagus, barley, wheat and garlic, which have associated prebiotic effects and act as a substrate for the microflora in the large intestine[5].
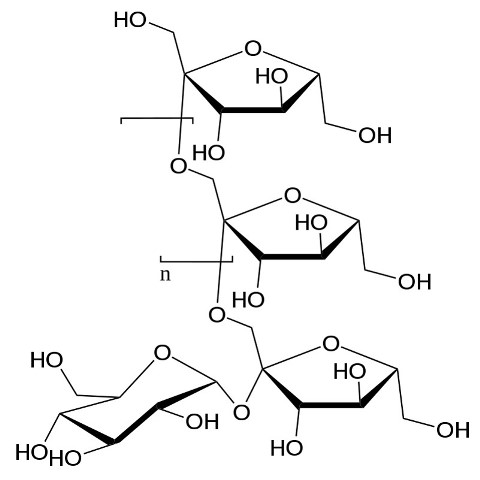
General structure of fructooligosaccharides. Source: Wikipedia.com
Polysaccharides, as their name suggests, are the result of the condensation of a large number of monosaccharides. It could be said that they are like polymers, in which their monomers (main constituents) are monosaccharide molecules linked repeatedly by glycosidic bonds[6]. For example, some of the most common polysaccharides are: cellulose, which would be a polymer of β-glucose formed by the union of more than 3000 glucose units, or starch, which would be similarly, a polymer of α-glucose. Other common polysaccharides are chitin and glycogen.
Starches and fibers
Starch is one of the most common carbohydrates in our diet. It is included in cereals (rice, corn and wheat) and tubers such as potatoes and cassava, and in foods processed from these, such as tortillas, bread and pasta.
On the other side, dietary fiber is composed of the part of plant tissue that resists digestion, since it is composed of polysaccharides such as cellulose and inulin and other non-saccharide compounds such as lignin.

Structures of two common polysaccharides. Source: image edited by @emiliomoron, contains public domain images: starch formula, cellulose formula, bread, dietary fiber.
The amazing thing to note is how, although cellulose and starch are polymers of the same monosaccharide, the differences in their structure give them completely different properties from a biochemical point of view; since, although both compounds are linear chains of glucose, starch contains alpha bonds while cellulose contains beta glycosidic bonds, so, while starch is degraded by enzymes and bacteria in the intestine, cellulose is not. This converts starches into energy sources, and although cellulose cannot be digested, it also has an important role, as its effect in relieving constipation and the relationship of fiber-rich diets in the control of obesity is well known[7].
To summarize
Carbohydrates are carbon compounds, mainly sugars and their derivatives, which have become an important part of our diet, being the main source of energy for our cells.
Monosaccharides are simple sugars such as glucose, fructose and disaccharides such as sucrose, because of their potent sweetness, are used as added sugar to our food. On the other hand, more complex carbohydrates (oligosaccharides and polysaccharides), are polymers of simple sugars and constitute an essential part of our diet as they are an important source of energy and promote intestinal health, which brings other health benefits.
But we must keep in mind that excesses are bad, our processed foods make it easier for us to consume large amounts of carbohydrates, which is transformed into excess energy that the cells do not consume, so they are transformed into fat as a way to store that energy, and this is related to overweight.
Well friends, I hope you liked the information and with it you have expanded your knowledge about this food group. See you next time!
References
- Wikipedia.com. Carbohydrates.
- Wikipedia.com. Galactosa.
- Wikipedia.com. Lactosa.
- Wikipedia.com. Oligosacáridos.
- Wikipadia.com. Fructooligosacárido.
- Wikipedia.com. Polisacáridos.
- Wikipedia.com. Fibra alimentaria.
Congratulations a really well put together post with a detailed explanation of each of the elements of carbohydrates. Thank you for deepening that knowledge and sharing it with us.
Thank you very much, I am glad you found the information well explained, I hope it has been very useful to learn more about carbohydrates.
Interessant hoe je uitlegt dat koolhydraten zo’n grote rol spelen in onze voeding, maar dat brede chemische veld kan soms best overweldigend zijn. Toen ik zelf na een lang stuk lezen even wilde ontspannen, kwam ik terecht bij gambloria , waar ze bonussen aanbieden speciaal voor spelers uit België. Een paar rondes op de Book of Dead-slot gaven me precies die mentale pauze die ik nodig had, en die kleine spanning zorgde ervoor dat ik later met meer focus terugkeerde naar de stof.