When it rains acid
There are numerous hair-raising facts about acid rain that have stirred up uncertainty about the droplets that are deemed hazardous by many. Although the effects of acid rain can not be understated, they are not the way they seem.
The picture of acid rain we often paint will be nothing short of a skin-melting drop of acids that would melt and destroy whatever it comes in contact with. But what if nothing can be further from the truth in this case? Well, you are just about to find out in this article.
What Exactly Is Acid Rain?
Acid rain is physically not different from the regular rain that comes with its season, except for impurities that are a result of atmospheric pollution that makes it unsafe for use and harmful to organisms and their environments.
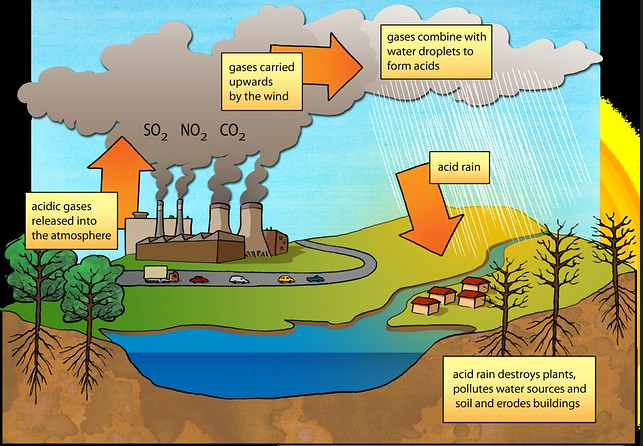
The term is relatively broad. It includes any form of precipitation (that could either be in a wet or dry form) that contains acidic components like sulfuric or nitric acid. This acidic precipitation can be in the form of rain, snow, fog, or hail. It might also interest you to know that even dust can be acidic too and it takes a spot in the category of acidic precipitation.
Why is it termed acidic? Will it burn the skin? Can it cause a piece of metal to rot? There are tons of questions and uncertainties triggered by the thought of 'acid rain'. It could frighten or boggle your mind but it is noteworthy that acid rain is not a downpour of raw concentrated acid. It is termed 'acid rain' because it contains some mixtures and components that make it a little acidic than normal. Pure water has a pH of 7. And the regular rainwater could have a pH of 6.0 or a little less. The pH of acid rain can be around 5.0 or 5.5. Some countries populated with cars and more industries can have acid rains with a pH that ranges between 4.0 to 4.4.
What Causes Acid Rain?
Several factors, man-made and natural, contribute to the downpour of acid rain. Some of these actions include volcanic eruptions, rotting vegetation, burning of fossil fuels, industrial activities, automobiles, and different power plants. These activities release some chemicals and components that can result in acid rain.
Chemicals such as sulfur dioxide (SO2) and nitrogen oxide (NO) are released into the atmosphere when fossil fuels are burnt. These chemicals can rise high to thousands of miles into the atmosphere where they react with oxygen and water as well as other components to create more acidic pollutants such as airborne nitric and sulfuric acids which later come down in the form of 'acid rain.
Effects Of Acid Rain
There are various ecological consequences of acid rain that pose a threat to agriculture and animals. They are mostly heavy on marine habitats like the lakes and streams, marshes, and ponds where aquatic wildlife can become toxic. Acid rains can over time lessen the pH level of an aquatic body and a pH less than 4.8 can make the water body inhabitable for aquatic life.
Acid rain is also known to take a toll on trees and forests. It is a renowned natural method of deforestation. It makes trees less immune to death by disease and wild insects, by eroding their leaves, destroying their barks, and attacking healthy growth. Forest damage is one of the effects of acid rain.
Acid rain is also capable of corroding and weathering buildings and metals such as steel, iron, copper, and bronze. Paints could wear off after a while of being exposed to acid rain. Marbles and limestone are easily damaged by acid rain as the calcium compounds in these materials react with the acidic components in the rain. Some statues and gravestones as well as eons-old monuments, and desolate buildings are being damaged by acid rain.
How Can We Curb Acid Rain And Its Adverse Effects?
The effects of acid rain on humans and their environments are not entirely out of control. While it is not feasible to prevent acid rain, there are ways to reduce its amount and automatically the effects.
Since nitrogen oxides (NOx) and sulfur dioxide (SO2) are the leading causes of acid rain and are produced from fossil fuel combustions, one of the most promising ways to put a cap on acid rain and its effects, and to ensure a safer environment is to build eco-friendly power plants and energy sources such as solar and wind power. Eco-friendly and renewable sources of energy produce fewer toxic emissions.
The effect of acid rain on aquatic bodies can be reduced with a process called 'linings. It is employed to fix the damage done on rivers, lakes and the likes, by acid rain. It balances the acidity on water surfaces and makes it a lot less hostile for aquatic life to survive. Although it is somewhat expensive and requires consistency for a lasting effect, it is used extensively to reduce serious acidity on water surfaces.

Conclusion
Acid rain might have adverse effects when it touches the earth's surface and its inhabitants but it is less terrifying than we think. It is just a thing that can be put in check if necessary measures are taken. Unless you live in the remotest part of a country, everyone must have witnessed acid rains before considering the level of industrialization in the world. However, since it can go physically undetected if it is not too acidic, we may not know. Those who live in heavily industrialized areas will experience it more due to the localization of the acidic oxides. However, since the oxides are gases, they eventually diffuse and become global.
References
- https://blog.arcadia.com/15-key-facts-and-statistics-about-acid-rain/
- https://www.softschools.com/facts/weather/acid_rain_facts/575/
- https://www.indiatoday.in/education-today/gk-current-affairs/story/acid-rain-320520-2016-04-28
- https://www.eartheclipse.com/environment/acid-rain-facts.html
- https://api.nationalgeographic.com/distribution/public/amp/environment/article/acid-rain
- https://www.epa.gov/acidrain/what-acid-rain
Acid rains are portrayed a lot worse, but as you have mentioned, it is not that threatening. !discovery 15
This post was shared and voted inside the discord by the curators team of discovery-it
Join our community! hive-193212
Discovery-it is also a Witness, vote for us here
Delegate to us for passive income. Check our 80% fee-back Program
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider supporting our funding proposal, approving our witness (@stem.witness) or delegating to the @stemsocial account (for some ROI).
Please consider using the STEMsocial app app and including @stemsocial as a beneficiary to get a stronger support.
I also only had in mind the way they are presented in the movies, it's still bad but it makes more sense now. Thank you!