Molecular swarm that modifies gold surfaces
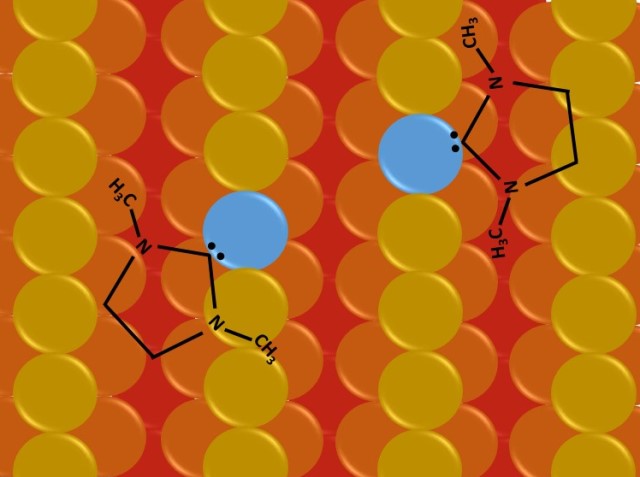
Molecules move atoms on a gold surface.
The surface of metals plays a key role in many technologically important chemical reactions, such as catalysis or sensor development. For example, in the large-scale production of many compounds, metal catalysts are used, and the surface characteristics of these catalysts determine whether and how a reaction takes place, which is why much research is focused on the development of new methods to adapt the structure of metal surfaces at the atomic level, as this provides a direct route for directing interfacial processes.
A team of chemists and physicists from the University of Münster has developed a method that allows to achieve this. The restructuring of the surface is carried out by individual molecules called N-heterocyclic carbenes, which are associated to reorganize the surface atom by atom. N-Heterocyclic carbenes, or NHCs have been widely studied as ligands of metal complexes precisely because they show a strong impact on the chemical and physical properties of the metal center, and more recently, because they show a strong interaction with the metal surface; the molecules absorbed on the surface generate ballbot type molecular species. In the study they found that a NHC molecule interacted with a single gold atom at a time, resulting in very ordered and stable monolayers. Using a computer simulation, it was possible to predict that at least two molecules of carbenes associate to carry out the restructuring, which takes place in a similar way to the closing of a zipper. As part of the study, the scientists were able to experimentally confirm this zipper-type mechanism by having carbene molecules work together on a gold surface to rearrange two rows of gold atoms into a single row. These results were published in the journal Angewandte Chemie International Edition, last August.
In previous studies this group of scientists had already demonstrated that these molecules of carbenes have a high mobility on a gold surface, however it had not been possible to demonstrate that there was any specific change in the metal surface by these molecules. Thanks to this last study the scientists did show that the structure of the surface can be modified and with great precision as a result of the cooperation of the carbene molecules. The scientists compared the association of these molecules as a molecular swarm, by the fact that they work together to achieve a change in the structure of the metal surface.
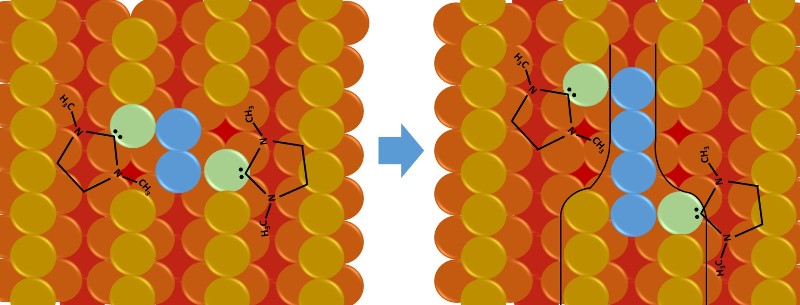
Like a zipper, the carbene molecules cooperate to convert two rows of atoms into one.
According to the results found, one NHC molecule extracts an atom and creates a defect on the surface, then another molecule reconfigures the created defect by moving a second atom, the coordinated interaction of the carbine-gold species along a specific direction results in a gradual restructuring of the surface by creating the Au-nanorods.
This procedure will allow the development of new materials with unique and very specific surface properties, which will undoubtedly influence the development of more efficient catalysts, electrodes and sensors. Currently, conventional tools such as presses and rollers are used to mold the surface of these elements, but with these procedures it is not possible to achieve such specific changes at the atomic level. We hope to see in the future how the help of nanomaterials will allow us to optimize surface chemical reactions, and thus develop more efficient and selective industrial processes.
Thanks for coming by to read friends, I hope you liked the information. See you next time.
Note: The images are of my authorship elaborated in PowerPoint, adapted of the described in the article of investigation.

Thank you for sharing this information on this type of technology that will change every day the way of thinking about science, as it shows us the structural part of the matter and very interesting how the future will favor us.
That's right my friend @newton666, technologies like this show us a structural part of matter that is difficult to see but that will undoubtedly benefit us in many areas
interesting post
Thanks!
This post has been manually curated by @bala41288 from Indiaunited community. Join us on our Discord Server.
Do you know that you can earn a passive income by delegating to @indiaunited. We share 80 % of the curation rewards with the delegators.
Here are some handy links for delegations: 100HP, 250HP, 500HP, 1000HP.
Read our latest announcement post to get more information.
Please contribute to the community by upvoting this comment and posts made by @indiaunited.
Thank you very much for your support