Control the oxidation of iron catalysts to obtain cheaper renewable fuels

Source: @emiliomoron.
A group of researchers at Washington State University have taken an important step toward converting vegetal materials into fuels more economically, and that is stabilizing the oxidation state of iron in the design of iron-based catalysts. The researchers appear to have found a way to prevent iron from oxidizing during the course of important chemical reactions needed to convert plant materials into fuels.
Headed by Yong Wang, the researchers reported their progress in the July 14th issue of the journal ACS Catalysis. According to the report, these researchers have been trying to find more efficient ways to produce fuels from renewable resources such as algae, but these biofuels tend to be more expensive and much less energy-intensive than fossil fuels.
The problem with using this type of material is that oxygen must be removed before it can be used, and one cheap metal that could do this is iron. But because iron also reacts with oxygen, it oxidizes while the reaction is taking place and stops working, so the process stops.
In this work the researchers prepared nitrogen-doped carbon compounds (NC) which they used as support for iron particles, and found that carbon materials whose structure was modified with nitrogen allowed more effective control of the oxidation state of iron through electronic interaction between iron and nitrogen particles. This interaction produces a decrease in iron particle size when the proportion of nitrogen in the carbonaceous support is increased and also leads to a weak affinity of oxygen for iron particles in the presence of the nitrogen-doped sites.
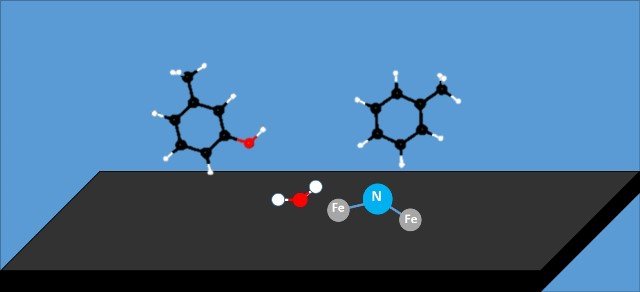
Representation of the surface of the catalyst. Source: image edited by @emiliomoron.
This lower affinity of oxygen for iron is related to a higher stability of the metal during longer periods of use of Fe/NC catalysts during the hydrodesoxygenation of lignin-derived m-cresol to form valuable aromatic products. An investigation of the surface composition of the nitrogen-doped samples showed that the constant rate of deactivation is closely related to the nitrogen content used to anchor the iron to the carbon structure of the support, indicating that the incorporation of nitrogen into the carbon skeleton plays a key role in stabilizing the deposited iron so that it interacts less with the oxygen while continuing to do the work necessary to remove the oxygen from the plant material.
The results were also confirmed computationally by electronic analysis of the iron-nitrogen complexes embedded in the graphene, demonstrating that nitrogen-containing sites in the catalyst skeleton protect Fe from oxidation compared to using undoped carbon as a carrier.
Now, although the work is promising, and marks the basis for the use of iron, an abundant and easily extractable element, in the development of catalysts for the conversion of biomass into fuel and other chemicals of interest; these catalysts will now need to be tested with actual feedstocks rather than the model molecules used for the study. Since the materials collected from the fields will be more complicated in their compositions and will present many impurities. But the research is a breakthrough on the way to finding more robust catalysts, based on low-cost, earth-abundant elements, that will make biomass conversion more cost-effective.
Thank you for reading me. Until the next post!

This post has been published previously in my other blog.
Greetings friend @emiliomoron, you share again an excellent sustainable alternative, in this case for obtaining renewable fuel, I find it very interesting that algae according to what I observe are plants that have multiple functions, because in agriculture also play an important role !
See you later my friend, have an excellent weekend ! 🤝
Thanks friend @amestyj. Certainly the algae are plants that are in the sight of many investigations, in the area of biofuels there is much interest for these.