New non-iridium based electrocatalyst for hydrogen production
Zero-emission hydrogen-powered vehicles are a reality, and in fact are a great alternative to electric vehicles, since they use fuel cells to generate the energy that drives the electric motor, so they do not require recharging periods connected to a plug socket.

Edited image, original from pxhere.com.
The problem is that obtaining hydrogen is not easy, it is usually obtained from fossil fuels, so the process ends up being just as polluting, and although you can resort to the process of electrolysis of water to separate the hydrogen from the molecule, but it requires energy and precious metal catalysts, such as platinum or iridium, which end up making this alternative very expensive.
That is why mass-marketing the hydrogen-based transportation system is not an easy task, but many researchers are working to make hydrogen more accessible, and one group of them may have taken an important step in that direction, as engineers at Rice University have developed a method that would make water electrolysis more economical to produce clean hydrogen fuels.
This group of scientists has substituted expensive metals, such as iridium, for ruthenium, a metal that is also among the precious ones but is much more abundant, using it as a catalyst in the positive electrode of a reactor where the separation of water into its constituent elements, hydrogen and oxygen, takes place.
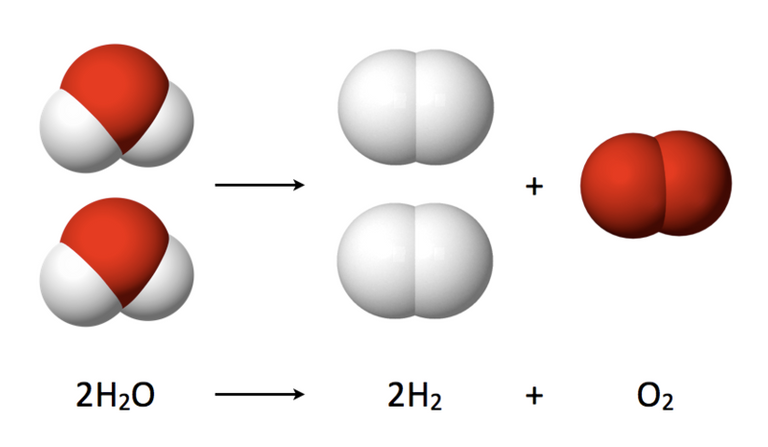
Source: Wikimedia.org.
It seems that by adding nickel to ruthenium dioxide, it was possible to develop a very effective anode catalyst to be used in the electrolysis of water, being active for thousands of hours under ambient reaction conditions.
Today there is great interest in the development of clean hydrogen, because once obtained it can be used in vehicle fuel cells, without releasing carbon into the atmosphere, neither in the production nor in the use in vehicles, since in these, hydrogen is subjected to the reverse process, mixing it with oxygen to produce water and electricity, so the only smoke released by a hydrogen vehicle is simply water vapor.
This catalyst could bring water electrolysis closer as a means of obtaining clean hydrogen, since ruthenium costs 8 times less than iridium, which could mean a 20 to 40% reduction in the cost of manufacturing useful devices for the market.
And if the electricity used in the reaction can be obtained from renewable energies, the most important limitation in the use of water electrolysis is precisely the catalyst, and so far the only practical option was iridium, since the electrolysis of water consists of oxygen and hydrogen evolution reactions by which the catalysts, when polarized, reorganize the molecule and reorganize the elements to release hydrogen and molecular oxygen, these reactions occur in an electrolytic cell, the release of hydrogen occurs at the cathode, and oxygen is produced at the anode, oxidizing the water to balance the charges.
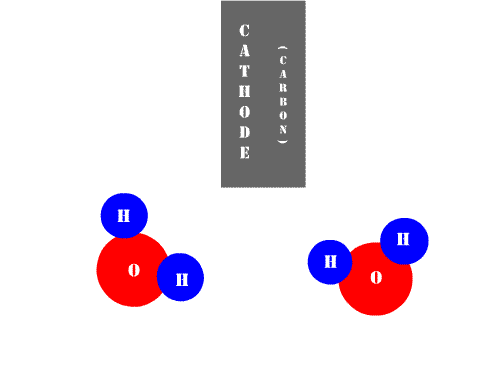
Source: Wikimedia.org.
The problem with this electrolytic system is that the anode is susceptible to corrosion, and when using transition metals such as iron, nickel or manganese they quickly oxidize and dissolve in the medium, so it is necessary to resort to expensive iridium.
But this research group tested ruthenium oxide, due to its already reported activity, but opted for doping it with nickel which had already been tested before, specifically they used RuO2 nanoparticles doped with nickel to make the anode of the cell, and managed to determine that this catalytic system remained active for more than 1,000 hours of reaction at a current density of 200 milliamperes per square centimeter, suffering negligible degradation. This represented a great improvement over the pure ruthenium oxides previously employed, which began to degrade appreciably after about 100 hours of activity.
This is undoubtedly a significant advance in the electrolysis of water to obtain hydrogen, since we cannot depend on a single catalyst to satisfy the amount of hydrogen needed today, much less if it is a scarce and expensive metal, we have to advance in the development of new efficient and sustainable catalysts.
References
Wu, ZY., Chen, FY., Li, B. et al. (2022). Non-iridium-based electrocatalyst for durable acidic oxygen evolution reaction in proton exchange membrane water electrolysis. Nat. Mater.
Wikipedia.org. Electrolysis of water
I don't understand much of this but I think this is interesting at least
Hi friend, thanks for stopping by to read, I'm glad you found the topic interesting.
You're welcome my friend
Thanks for your contribution to the STEMsocial community. Feel free to join us on discord to get to know the rest of us!
Please consider delegating to the @stemsocial account (85% of the curation rewards are returned).
You may also include @stemsocial as a beneficiary of the rewards of this post to get a stronger support.
Intersting advances in the hydrogen research
!1UP
There are great advances, let's hope to see methods like this applied soon.
You have received a 1UP from @gwajnberg!
@stem-curator
And they will bring !PIZZA 🍕.
Learn more about our delegation service to earn daily rewards. Join the Cartel on Discord.
I gifted $PIZZA slices here:
@curation-cartel(20/20) tipped @emiliomoron (x1)
Join us in Discord!
A very innovative proposal in environmental care.