Formic acid as a fuel to mitigate climate change
We are all aware of the relationship between greenhouse gas emissions and global warming, with CO2 being the gas emitted in the highest concentration and therefore the one that bears the greatest responsibility for this problem. But we are aware that today, combustion engines are also responsible for moving the economy and society, something that creates some dependence, and although progress has been made in the electrification of transportation, there is still a long way to go, so improving current technologies could be the fastest way to mitigate our impact on the environment.
And an interesting way to obtain energy that I have come across, and that could be a sustainable way to improve the operation of our vehicles, is through formic acid, an acid that we can find in nature as it is the poison injected by some ants and bees when they sting.

Formic acid can be very useful as a source of energy. Source: Image edited in powerpoint, original from pxhere.com.
And is that, with the emergence of the first hydrogen fuel cells using formic acid and later cells that directly use this acid, perhaps we are witnessing a new way of obtaining energy, which even prompted the development of the first bus that is powered by formic acid. This is a vehicle developed by Team Fast (Formic Acid Sustainable Transportation), a group of students from the Eindhoven University of Technology, a small vehicle they dubbed "Junior" was their proof of concept, a small vehicle that was able to convert formic acid into electricity.

Prototype driven by formic acid. Fuente: Wikimedia.org.
Formic acid, HCOOH, also called methanoic acid, is an organic substance possessing only one carbon atom, and is therefore the simplest of the organic acids, and this acid can function as a fuel for hydrogen cells in two ways.
It can be used to obtain hydrogen, which is the gas that essentially powers the cells, by catalytically reforming the formic acid to obtain hydrogen and CO2 . The advantage of this method is that the acid can be stored in liquid form, which is less dangerous than storing gaseous hydrogen to feed the fuel cells.
It can be used directly, which eliminates the need for the reforming stage, but is combined directly inside the cell with oxygen to produce H2 O and CO2 .
This way of storing and supplying hydrogen is simpler and less dangerous than storing hydrogen to power fuel cells, but it makes no sense to develop a technology that does not solve the problem of carbon dioxide emissions.
The ideal would be to maintain a zero balance in emissions, that is, to find a way to convert CO2 from the atmosphere into formic acid, which in turn is used as fuel releasing the same amount of CO2 , so no more carbon is incorporated into the environment.
The solution, direct hydrogenation of CO2 to formic acid
Formic acid is not only a valuable product for the chemical industry, but its decomposition into CO2 and H2 allows us to use it as a hydrogen storage element, but the best thing is that this transformation can be reversible, which gives us the possibility to regenerate formic acid.
But while this is possible, it requires complicated and energy-intensive refining processes, leaving us back at square one.
However, a team of researchers at the University of Science and Technology of China appears to have found a more efficient and environmentally friendly way to transform CO2 from the atmosphere into formic acid.
In a paper published in the journal Nature Nanotechnology, published last December, the research team claimed to have developed a system that uses renewable electricity to directly convert carbon dioxide into pure formic acid.
The research group has developed a copper-based catalyst alloyed with a single lead atom (Pb1Cu), with which they have succeeded in converting CO2 to formate with high selectivity (approximately 96%) in an electrochemical reaction system based on a solid-state electrolyte reactor. The laboratory-scale system performed continuous production of 8 L of pure 0.1 M formic acid solution.
Hopefully, research like this will promote the industrial recycling of CO2 using renewable electricity, which is vitally important to drive the circular CO2 economy.
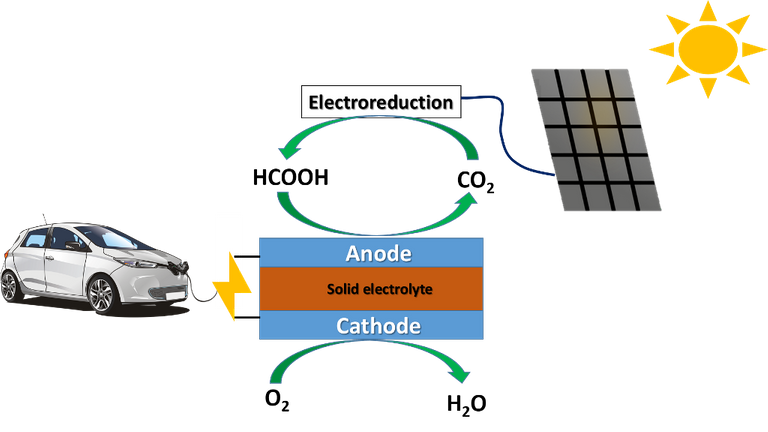
Recycling carbon dioxide to produce fuels will maintain a balance in emissions. Source: image elaborated in Powerpoint, contains public domain image.
In this way we would not be adding more carbon dioxide to the atmosphere, we would be reusing the carbon dioxide already being produced by industries and combustion vehicles, while paving the way for the total electrification of the means of transport.
Thanks for coming by to read friends, I hope you liked the information. See you next time.
Great one. I guess this will be more economical and help the degrading climate
It may not be as economical until it is industrialized, but it is certainly an alternative to maintain a balance in emissions.
Being able to balance emissions is a great advantage . It will make the world a safer and better place