Mitigating global warming with sunlight
As we know at present there is a growing concern about global warming, some of the most talked about actions to mitigate this phenomenon is to reduce our carbon dioxide emissions as provided in the Paris Agreement, with the aim of limiting the increase in the average temperature of the planet.

We can use the sun's energy to mitigate climate change in different ways. Source: publicdomainpictures.net.
In this sense, different strategies have been addressed to reduce CO2 emissions to the atmosphere, the most accepted being the change of energy generation systems based on fossil fuels for more environmentally friendly means and renewable sources, such as the use of wind and solar energy. This is in order to avoid producing more greenhouse gases.
And although the current major effort is focused on developing alternative technologies and using renewable energy sources, there is still the problem of reducing the current levels of CO2 in the atmosphere; and in this regard, much has been said about various methods to capture and store CO2, such as its capture and mineralization in solid absorbents or its storage in the seabed. All these techniques, although they offer a solution to reduce the current levels of CO2 in the atmosphere, also offer several technical difficulties and leave latent the possibility that all the captured CO2 can return to the atmosphere if its place of deposit is disturbed by some natural event.
But there are other ways to remove CO2 from the atmosphere, and recent efforts seem to come from the use of photocatalysts, compounds capable of acting as semiconductors when excited by some energy source, such as sunlight. In this sense, instead of capturing and storing CO2, the idea is to convert it into other compounds, similar to the way plants harness it through photosynthesis.

The idea is to mimic the reactions carried out by plants through photosynthesis. Source: pxhere.com.
And although this idea has been on the table for a long time, the photocatalysts so far designed have not been very efficient in carrying out this process in large volumes, but this could take a turn, as a group of researchers at the Nagoya Institute of Technology in Japan are exploring a photocatalyst that is much more efficient than those known to date.
In their research published in the journal Nature, the researchers have focused on the use of silver iodate, a compound of great interest since it has been useful for CO2 reduction but had not been viable because it required more energy than that supplied by sunlight to carry out the reaction; therefore, these scientists presented a novel system that incorporates the use of carbon nanotubes in combination with silver iodide to improve the efficiency of the catalytic system and convert CO2 into carbon monoxide.
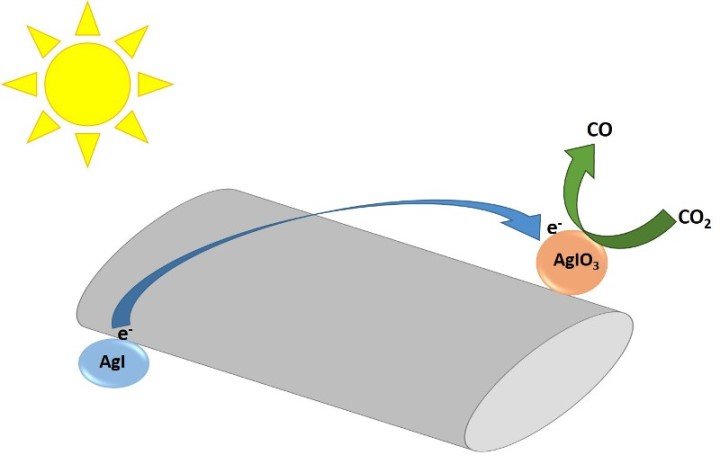
Mechanism of the photocatalytic system. The photoexcited electron from silver iodide (AgI) travels through the carbon nanotube to silver iodate (AgIO3), where carbon dioxide is reduced to carbon monoxide (CO). Source: image elaborated in PowerPoint.
By incorporating the carbon nanotubes, the dispersion of the composite was easily coated with a thin film polymer to produce flexible photocatalytic electrodes, so that they can be used in a variety of applications. With this, the reduction of industrial CO2 and or atmospheric CO2 emissions can be addressed through an easy-to-use, renewable energy-based solution.
Hopefully, the system can provide the necessary results for scalability and in the future companies that still rely on fossil fuels to generate energy can use such a catalytic system to reduce their emissions, or we can have systems based on this catalyst on the surface of large buildings to reduce atmospheric CO2 from human activities.
Thanks for coming by to read friends, I hope you liked the information. See you next time.