COVID-19 vaccination - is it worth it?
Dear Hivers,
first I just wanted to comment on the post from @perceive about the side effects seen in the ongoing COVID-19 vaccine trials. But then the comment got longer and longer, so here are my 2c on this topic.
Actually I work in drug development, in a pharma company, so you can believe me that I know what I am talking about. And I can tell you that those chills and fever described by @perceive are quite normal. They are of course annoying, but relatively harmless. Also that a Brazilian trial subject died recently is tragic, but not worrying, as it turned out he was in the (placebo-treated) control group.
However, what is not harmless are the longterm side effect that won´t show up quickly and obviously!
Our immune system is really complex and far from being fully understood. But one thing we know: it doesn´t forget things. Such harsh immediate reactions described will have in addition long term triggering effects which nobody can predict for sure. Each human has a different immune system and capability, depending on many variables, e.g. overall health state and age, but also healthy diet, low level of pollution, a well functional relationship, lack of essential troubles,.. The relatively new area of psychoneuroimmunology deals with the delicate interactions between psychological processes and and our immune system. To summarize, we can´t know for sure, in what way a boosting of our immune system by a vaccine shot triggers unwanted immune reactions. Take the H1N1 (swine flu) vaccine Pandemrix as an example.
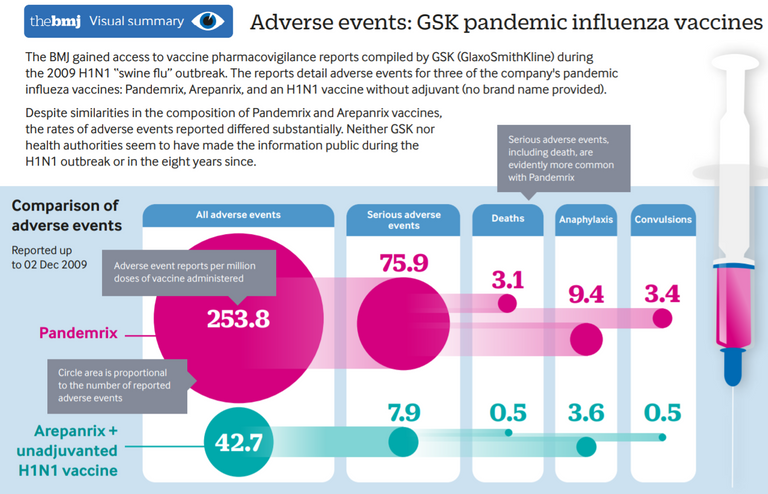
Pandemrix was known to have much more side effects than comparative drugs even before the narcolepsy side effect was known (source)
More than 1000 children and youths in Europe had developed the very uncommon but devastating sleep disorder narcolepsy after having been vaccinated for H1N1 with Pandemrix which contains a specific adjuvant. Other H1N1 vaccines not having this adjuvant did not lead to increased narcolepsy cases. Narcolepsy is today known to be an auto immune disease. We can safely assume that with every vaccine there is - at the time the drug is used - an unknown percentage of such unwanted long term effects. So the rationale to take this risk should be better very solid!
Generally, autoimmune reactions and diseases are surging worldwide since years. So much that the pharma companies can´t fill the demand of certain drugs which are fighting these diseases, despite being partly super-expensive! A company would for sure not want to lose business by not supplying enough product, but exactly this is the case.
Why?
Pollution & unhealthy processed food and life style for sure are main drivers, but auto immune reactions from vaccine adjuvants like mercury or aluminum play a role as well, Pandemrix is only one example of several! There is even a new syndrome called "Autoimmune Syndrome Induced by Adjuvants" (ASIA), but it is until today not clear in how far this is really clinically relevant (1) and some authors conclude that current studies do not support the existence of ASIA. But to evaluate the significance of ASIA is not the point of this post. It is a whole story on its own.
The point is that due to these not well known risks only do a vaccination when the benefit outweighs the risks. Until you not belong to a high-risk group (immune depletion, cancer, cardiovasc. diseases, high age) this is not the case for COVID-19, because new epidemiologic data (e.g. (2)) show that the mortality rate of COVID-19 is not more than that of a severe flu, especially in people <70y old.
You might argue that there are drugs out there that have much more side effects than a vaccine, like used in cancer. As you know they are in part almost as deadly as the cancer itself. But in order to save a few weeks time of life, people do take them. We cannot blame them or the treating physicians, until we are in the same situation, facing close-by death. At least I am not sure if I would have the guts and say no to such drugs. I just mentioned it, because it is the severity of the disease or condition which decides what side effects one should be willing to accept. In that extreme case of a deadly disease it might be totally acceptable to take drugs which are very troubling. On the contrary, if you suffer from a tiny headache, you would never take a pill from which you will lose your hair, right?
And same is with COVID-19. The more it turns out that this is a disease like a flu, the less risks we should be accepting in the prevention of it. Especially politicians should be overly careful about the risks when they start MASS VACCINATION programs, because then even very rare side effect will accumulate in the treated population. You have to know about the rarity definitions which are worldwide standardized:
| Category | Frequency | In % |
|---|---|---|
| Very common | ≥1/10 | ≥10% |
| Common (frequent) | ≥1/100 and <1/10 | ≥1% and <10% |
| Uncommon (infrequent) | ≥1/1000 and <1/100 | ≥0.1% and <1% |
| Rare | ≥1/10,000 and <1/1000 | ≥0.01% and <0.1% |
| Very rare | <1/10,000 | <0.01% |
If you dose several hundred Mio people (as some politicians and profiteers wish), even very rare side effects add up. Let´s suppose 500 mio. sheeple get vaccinated (not entirely unrealistic) and let´s further suppose that the drug has a very rare, but also very serious side effect at a frequency of 1:10.000 (0,01%) - that´s quite underestimated given what was said above. Then on average 50.000 people will get this side effect! We are speaking about otherwise healthy people most of which never would get harmed from this flu-like virus but from the vaccine for sure around 50.000 people will be seriously harmed per 500 mio. vaccinated. And on top of it, the protection rate from that vaccination won´t be 100%. This again decreases the risk-benefit ratio significantly. Let´s suppose optimistically that 50% of the vaccinated people are protected from COVID-19 (assuming typical protection rates from flu vaccines and a CDC-estimate (3)), then you need to vaccinate 1000 mio. people to protect 500 mio, and will have 100.000 side effect victims! And for a still rare event with a probability of 1:1000? Then it will be already 1 Mio. adverse events in the vaccinated population! A price no responsible decision maker should be willing to pay! Well it´s rather the people who will pay the price at the end! And don´t forget, there has never been a Coronavirus vaccine. It is not at all clear that a 50% protection rate is even feasible! The clinical development programs currently ongoing hope for 60% but are satisfied, if the protection rate is >30% (4).
But how many deaths will be prevented by the vaccine? It largely depends on the efficacy of the drug (which nobody knows so far) and who gets the vaccine. Vaccinating 500 Mio. people <65y would be a desaster, as this group is hardly affected by SARS-Cov-2. Clearly, only risk groups should be vaccinated (like it is also recommended for flu shots). But here the problem is that especially in very old people and people with immune defects, a vaccination is often not effective, because you need a strong, active immune system to have the optimal triggering effect by the vaccine. Tragically, especially the groups which would profit most, are the ones where the vaccination is least successful. Besides, any vaccine currently in development for sure won´t have this vulnerable group of people in focus of their clinical trials. In case of the ongoing Moderna Phase 3 trial, patients with "Immunosuppressive or immunodeficient state" are explicitly excluded from trial participation ((4), page 45)! All this doesn´t improve the benefit-risk ratio in this group, which brings me to the conclusion that for nobody (both low- and high-risk groups) it is worth to get a COVID-19 vaccination. But hard facts we will know only years from now.
Did I mention that those very rare but potentially serious side effect are nearly IMPOSSIBLE to detect upfront in those rushed clinical trials we are seeing now? To detect a side-effect of 1:10000 that occurs within 5 years, you would need to observe vaccinated patients of at least the same number for 5 years (and even this is no guarantee you will spot it). No way, that the Authorities worldwide will wait until then before declaring the vaccine as safe.
But there is even more!
Some of the more than 150(!) COVID-19 vaccines in clinical development (for a summary see here) are even of a completely new category, gene-based or mRNA vaccines (5) (made by companies like Moderna and Curevac), which were so far never licensed in humans and where one should be especially cautious. Why? By inserting viral mRNA into the cells, they trick the body into producing some of the viral proteins itself. I for sure don´t want that my cells produce viral proteins unless it is fully explained in what ways this triggers the immune system and until long term safety follow up studies are published! Who could ensure that such a radical new immune stimulation can´t induce in a subset of recipients a much-feared cytokine storm or cancer inductions years later?
Despite the risks, they are loved by the companies and the mass vaccination promoters because they are way cheaper and quicker to produce than traditional vaccines (5).
And to make things worse, in a recent Forbes article (6) it is revealed how pathetic the trial design of several ongoing Phase 3 vaccine trials from Moderna, Pfizer, Astra Zeneca and J&J is!
As an example, in the Moderna trial COVID-19 cases are defined by these vague criteria, e.g. a positive PCR-test plus a few symptoms, nothing else, particulary no virus isolation from blood and no ruling out of another infection like flu.
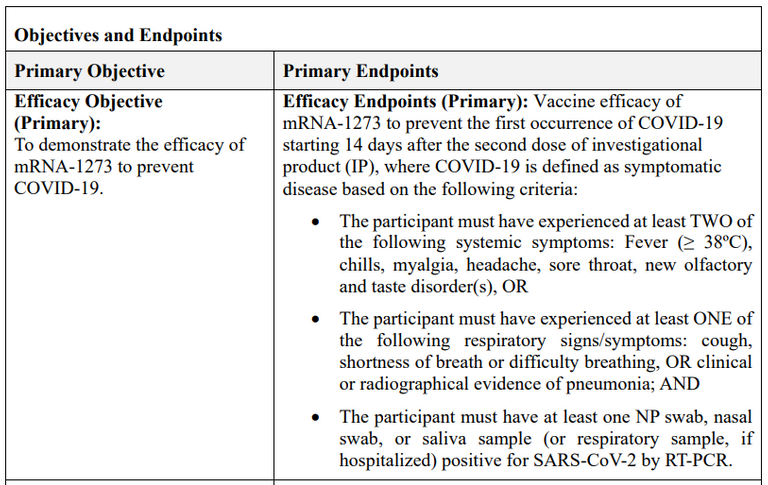
COVID-19 definition in the Moderna Ph3 trial (source)
Also, already after only 53 such COVID-19 events a first interim analysis is done, and as long as 13 or less of those 53 cases were in the vaccinated group compared to 40 or more in the unvaccinated control group (which is of same size), this is considered a success already! Pfizer is even more generous in its success requirements. Their interim analysis starts after 32 vaccine recipients develop COVID-19 symptoms, with a success margin of 7 or less in the vaccinated compared to 25 or more in the control group. It is not even aimed (nor realistic) to totally prevent SARS-Cov-2 by the vaccination.
If no "success" is showable after 53 events, they check again after 106 events, and if then still no evidence, they try again after 152 events:
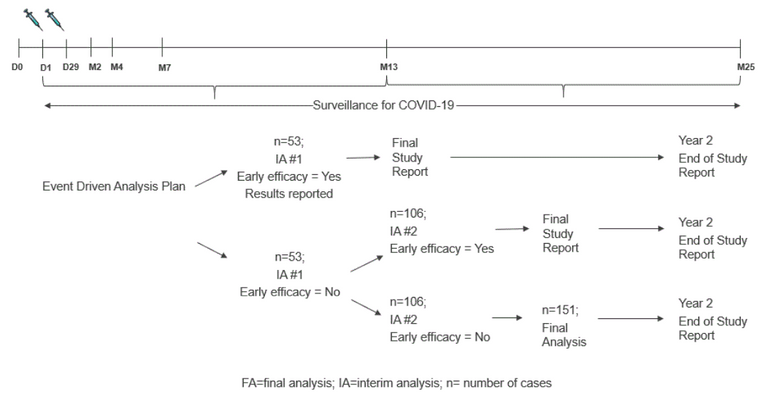
Event-driven analysis plan in the Moderna Ph3 trial (source)
If the "early efficacy" is shown, they will already publish the results and go with that (and without any safety follow up data!) to the Health Authorities, to get a speedy priority review and approval of the drug. Of course the study continues to follow up all vaccinated subjects for 2 years (why not longer??), but by then the vaccine is probably given already to ten thousand people or more. And remember, in case of Moderna, this is a mRNA vaccine that was never tried out in humans!! Extremely scary!
In fact, the vaccines will be so "safe" that the vaccine makers won´t cover any costs due to side-effects of their COVID-19 shots but will be indemnified in Europe and the USA (e.g. AstraZeneca or Sanofi have fixed such deals with the EU recently (7))! Don´t they trust in their own medications?? It is unacceptable that the taxpayers will have to pay those costs!
SUMMARY:
It is highly doubtable that the COVID-19 vaccine will be safe enough to take the risks of the vaccination, simply because there is not enough time to wait until very infrequent adverse effects will show up in clinical trials.
The overall mild disease COVID-19 (median infection fatality rate is 0,05% in people <70 (2)) does not require taking such risks. If you as a politician want to reduce deaths from COVID-19, it is much better to protect the high-risk group (very old and/or vulnerable people) and having them stay at home and minimize social contacts, then to risk 100.000s of serious adverse reactions from mass vaccination programs, let alone all the other, not only economic costs from the panic spread like depressions, missed focus on other diseases,...
Sources:
(1) https://pubmed.ncbi.nlm.nih.gov/25794485/
(2) https://www.who.int/bulletin/online_first/BLT.20.265892.pdf
(3) https://www.cdc.gov/flu/vaccines-work/vaccineeffect.htm
(4) https://www.modernatx.com/sites/default/files/mRNA-1273-P301-Protocol.pdf
(5) https://horizon-magazine.eu/article/five-things-you-need-know-about-mrna-vaccines.html
(6) https://www.forbes.com/sites/williamhaseltine/2020/09/23/covid-19-vaccine-protocols-reveal-that-trials-are-designed-to-succeed/#229516025247
(7) https://www.indiatoday.in/science/story/covid-19-vaccine-makers-see-eu-shield-against-side-effect-claims-1724398-2020-09-23

PS: The #leofinance tag was added because covid-19 and the way it is acted upon impacts the economy of our world as we know it.
https://twitter.com/ArtOfTheMystic/status/1320424301464604672
Very informative.
Posted Using LeoFinance Beta
Thanks!
I am currently reading the book by Clemens Arvay "Wir können es besser". He addresses the issues of vaccines on YouTube, but here is his website, with all the books and links: https://www.clemensarvay.com/
Thank you for the information
Important contribution!
Tatsächlich? Ist das neu, oder hast du das schon mal gesagt?
Nein, schon seit etlichen Jahren. Da und dort werde ich es sicher schon mal anklingen haben lassen. Hab´ ja schon einige posts zum Thema geschrieben, z.B. hier.
Interessant. Ja, stimmt du hast diese Themen schon paar mal behandelt, allerdings hielt ich dich einfach nur für belesen.
Es ist gut, dass du aus der Branche bist und dadurch Ahnung hast/bekommst.
Vor allem praktisch, wenn ich mal was in die Richtung schreibe.. dann kannst du "ne ne ne, so stimmt das aber nicht" sagen.
Ohne dich zu sehr strapazieren zu wollen, hast du diesen Artikel vom Rubikon schon gesehen?
https://www.rubikon.news/artikel/die-medikamenten-tragodie
Danke!
Ein exzellenter Artikel, gut recherchiert und sollte in keiner Sammlung fehlen. Danke fürs Teilen.
DR THOMAS COWAN SYSTEMATICALLY DECIMATES THE COVID-19 LIE!

https://www.bitchute.com/video/TEThRUAWIxR1
Many thanks, I think you have made some important points here.
After what I have researched and read and heard over the last years, and in particular over the last months, I have come to the conclusion anyway that there has never been any really long-term and detailed documentation of vaccination damage in the history of vaccination and that the entire population has always been considered a testing ground.
It is extremely difficult to prove monocausality, in both directions.
I always think of the example of the death of bees, which have been said to be dying en masse and which environmentalists have suspected to be caused by the pesticides sprayed on fruit plants. But the companies that produce the pesticides have an easy job of insisting that there must be unconditional proof that the pesticides are harmful and that there are no other causes of bee mortality. To the extent that laboratory tests seek to prove this, it can be argued that they are just laboratory tests and not field research. However, if you do field research, you will find that you are actually dealing with such multiple factors that it is difficult to provide monocausal proof.
So we are dealing with the same problem again and again. Where laboratory research is carried out and then claims to have found something useful and effective, it may be brought to market if sufficient scientific consensus has been reached. Without long-term field research results; because what does long-term actually mean?
But now there is a difference between whether something is brought actively into play by technology. Or whether it is a phenomenon not influenced by man. For example, flu is one such phenomenon that we have been living with for a long time without panicking about it.
You rely on the fact that you would notice it if there was a sudden excess mortality or serious problems. At least that is what we understood by normality. We let events happen and then we act on them locally and with reason.
To live in a world where everyone is always suspicious, constantly monitoring each other, trying to avoid any harm, what is the gain? Certainly not a relaxed and spontaneous life. Where getting ill and dying is declared a taboo, the prolongation of this lifestyle is, in my opinion, not a quality but rather a torture.
I have never had the idea of blaming anyone for getting the flu. Nor do I want to start doing so. We harm each other in a social and mental way when we insist on this rigid monitoring and distrust.
Greetings to you.
Man merkt, dass Du hier in Deinem Element warst.
Was fehlt ist, ein Hinweis auf die Gefahr von antibody dependent enhacement im Falle einer Impfung, so wie es bei SARS-CoV1 Viren bereits gefunden wurde.
Es besteht also die Möglichkeit, dass ein Impfstoff das Risiko an COVID-19 zu versterben schlussendlich sogar noch erhöht. Das wäre dann die ultimative Katastrophe.
So many things to disagree on, but referencing Ioannidis? The guy even predicted max 40.000 deaths in the US by end of the year, WITHOUT measures. We have 200k and counting. Also I hear your concerns about the long term effects of the vaccines. How about the long term effects of the new corona? And there's plenty of research indicating that this is gonna be a big problem in the future :)
"Here is an effort to dissect why I was so wrong. Behaving like an expert (i.e. a fool) was clearly the main reason. " source
I wouldn't trust a fool.
It is normal that, as new information is available, the models change. The latest publication from Joannidis is based on a much more complete set of data, so I don´t care about his early predictions.
And when you say 200K and counting, please don´t forget that any death falls into this number, even terminal cases of cancer of patients who happen to get tested positive. Especially in the US where there is a financial benefit for having a Covid death vs. a non-Covid death.
Long term effects of Corona? Which ones?